12. Animals and Blood Transfusion
Doctors misled for over 200 years:
History shows that a dependence on animal research delayed the introduction of blood transfusion by over 200 years.
NAVS Leaflet, Ever had a blood transfusion?
The assertion that animal experiments delayed the development of blood transfusion derives from the superficial and inaccurate accounts found in animal rights literature (see for example Ref. 1, page 157; Ref. 2 page 220). A brief review of primary sources reveals that animal experiments were crucial to the development of a) the concept of the benefit of blood transfusion, b) techniques for carrying out transfusion and c) the preservation of incoagulable blood and thus to the establishment of blood banks.
The function of the heart and the details of the circulation of the blood through arteries and veins were, of course, demonstrated by William Harvey in 1628. Harvey used about 50 species to elucidate the nature of the circulation. He studied, by means of a magnifying glass, the motion of the heart in “a small shrimp” found in the Thames (probably a species of mysid (3)). He obtained clear proof that the heart pumped blood via a closed circulation through arteries to the veins by simple experiments, using ligatures, on the slowly beating hearts of cold-blooded animals. In warm-blooded animals he could “neither rightly perceive at first when the systole and diastole took place, nor when and where dilatation and contraction occurred by reason of the rapidity of the motion, which in many animals is accomplished in the twinkling of an eye, coming and going like a flash of lightning” (4). Experiments on sheep, deer and dog demonstrated that the heart pumped in unit time a larger quantity of blood than is found in the whole body – thus clearly demonstrating a circulatory system.
By the early 17th century therefore, the concept that blood was pumped via the arteries to the organs was well established. To some observers at least, the blood began to be perceived not only as “the vehicle of the soul with the secrets of individuality” (5), but as a transporting system. It was therefore no surprise that experimenters should endeavour to infuse medicaments into the circulating blood of a living animal. Christopher Wren and Robert Boyle, in 1656, inserted a quill attached to a bladder (in later experiments, a syringe) into a superficial vein of a dog and successfully injected first opium, then the emetic, antimony oxide (5).
Early Transfusion Experiments
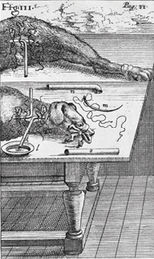
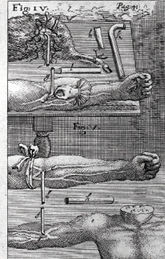
Figs. 12.1a and 12.1b Engraving showing transfusion in the neck and leg of a dog, from animal to man, and from man to man, by J. S. Elsholtz, 1667. Wellcome Library, London, CC BY.
This experiment doubtless led to attempts to transfuse blood from one animal to another. The earliest, well-authenticated account of the transfusion of blood from one dog to a second is that of Richard Lower, who in February 1665 used quills (and later a silver tube) to transfer blood from the carotid artery of one dog to the jugular vein of a second (6) (Fig. 12.1a). A later demonstration of the technique is described in Pepys’s diary of November 14th 1666, where the recipient dog was described as “very well, and likely to do well.” The diary also records subsequent discussion, which included musings as to the effect of transferring blood of a Quaker to an Archbishop. Boyle also queried whether the transfused dog would still recognise his master after receiving the strange blood (6). Thus although animal experiments had demonstrated the feasibility of transfusion, many still regarded the blood simply as the vehicle of the soul. The potential of the technique was clearly not appreciated.
The first person to transfer animal blood to a human was the French philosopher and mathematician Denis (spelt Denys in some accounts). With the help of the surgeon Emmerez Denis in 1666 allegedly transfered 9 ounces of blood from the carotid artery of a lamb into a 15 year old youth, moribund following excessive venesections to alleviate an obscure fever (Fig. 12.1b). Apparently, astonishing improvement resulted. Denis performed the operation on a further three subjects with no untoward effect, although the artery to vein anastomoses, with the chance of coagulation, made estimations of the amounts transfused dubious (Fig. 12.2).

Fig. 12.2 Attempt at blood transfusion from lamb to man, depicted in an illustration dating from 1705. Wellcome Library, London, CC BY.
His last patient was a man “with an inveterate phrensy, occasioned by a disgrace he had received in some amours,” presumably a gentle euphemism for neurosyphilis. Denis considered that calf’s blood “by its mildness and freshness might allay the heat and ebulition of his blood” (7). The patient apparently tolerated two transfusions with some benefit upon his mental state, although the second was followed by pain in the kidneys and the production of black urine. At the behest of the patient’s wife Denis attempted a third transfusion after which the patient died. Denis had enemies prominent in the Faculty of Medicine of Paris who were implacably opposed to transfusion of animal blood into man. He was charged with murder but eventually exonerated after counter charges that the patient had been poisoned by his wife. Nevertheless experiments on transfusion of blood into humans were prohibited by an edict of the French Parliament.
This official ban on transfusion in France is proposed by opponents of animal experiments as an argument that such techniques thus “delayed the practical availability of blood transfusions and led directly to the deaths of patients” (8). However, if transfusions between different species had been investigated before the transfusion of animal blood to man, the incompatibility of bloods of different animal species would have been established in the 17th century instead of 150 years later.
The efforts of 17th century opponents of transfusion to stigmatise the technique as evil and against nature probably worked for the best. Medicine and technology were not sufficiently advanced for doctors to use transfusion effectively. Knowledge of the function of the blood, of sepsis, of immunology and of clotting mechanisms were minimal or non-existent. Methods for transferring coagulable blood between individuals, without the benefit of materials available today, were fraught with problems and danger. It was therefore not surprising that during the 18th century references to transfusion were rare, and those described were irrational (in 1792 for example, Russell claimed to cure a child of rabies by injection of lamb’s blood (6)).
Scientific Reasons for Transfusion
The gradual accumulation of physiological and pathological knowledge changed the perspective with which transfusion was viewed. Rosa and Scarpa (1788) recommended transfusion as a treatment for anaemia, and in 1796, Erasmus Darwin (grandfather of Charles) advocated tranfusion of blood in cancer of the oesophagus and other conditions resulting in inadequate nutrition. Darwin suggested transfusion could be effected via goose quills connected by a piece of chicken gut (6). There is no evidence that he attempted transfusion using this technique, but it is a salutary reminder of the primitive nature of the tools available for a complicated procedure.
Transfusion for Haemorrhage
Provost and Dumas in 1821 showed that animals haemorrhaged to the point of death could be revived by transfusion of blood, but not serum or water warmed to 38oC. Blood from animals of other species was not effective, since the animals appeared to survive but succumbed within a few days. Provost and Dumas did not attempt transfusion in humans, since they considered the basic knowledge of blood and its functions too rudimentary and, since the technique was too celebrated, it had already “been abused in an ignorant and barbarous century” (6).
Blood transfusion in humans was established as a sound scientific and clinical procedure by James Blundell (1790-1877), a lecturer in physiology and midwifery at the United Hospitals of St Thomas and Guy. Blundell was moved by the many deaths he had witnessed in patients with post partum haemorrhage. Even when bleeding had been suspended, frequently the patients had lost so much blood that one could do nothing but observe them sinking until death followed within 2-4 hours. In such cases, argued Blundell, “there is a fit opportunity for trying the operation of transfusion” (9).
In his extensive monograph (9) Blundell described his experiments in dogs that established that death from haemorrhage could be prevented by transfusion of blood from the same species, even if vital signs had been lost. Recovery occurred even if the volume of the blood transfused was a fraction of that lost (even just 20%). Transfusion of blood from another species was not effective, but venous blood was as effective as arterial blood, even if its transfusion was delayed or if it was passed through a syringe.
After his experiments in animals, Blundell took the giant step of attempting transfusion of human blood to patients with severe haemorrhage. He performed the operation 11 times, at first only as a last resort in patients who were clearly irrecoverable. As experience with the technique was gained it was used in appropriate, seriously ill patients quite successfully. A typical case was reported in The Lancet in 1828 (10). One and a half hours after delivery a woman collapsed with extreme prostration, “blanched and perfectly bloodless in appearance.” It transpired she had been bleeding freely into the uterus, unknown to the physicians. Stimulants (brandy and port wine) were freely given to no avail. Blundell transfused 8 ounces of blood, and the patient “rallied and became in every respect much better.” The patient made a full recovery and later commented that she had “felt as if life were infused into her body.”
In view of the obvious difficulties associated with supplying blood by the direct connection of the donor’s artery to the recipient’s vein, Blundell developed apparatus that obviated the need to cannulate the vessel of the donor. Venous blood was allowed to collect into reservoirs from whence it was pumped by syringe or allowed to flow under gravity (Blundell’s “impellor” and “gravitator”; Fig. 12.3a; Fig. 12.3b).
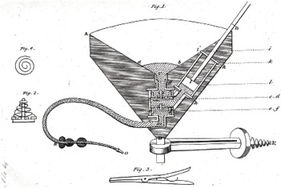
Fig. 12.3a Drawing of Blundell’s impellor, which allowed venous blood to be collected in reservoirs before being pumped or allowed to flow under gravity to the recipient. (From Ref. 9). Wellcome Library, London, CC BY.

Fig. 12.3b Blundell’s apparatus in use. From J. Blundell (1828). ‘Observations on the transfusion of blood’, The Lancet, 2, 321. Wellcome Library, London, CC BY.
Blood transfusion was thus established as a respectable and valuable procedure. It was not however commonly practised, but used as a last resort only. Some deaths still occurred even with human blood, but the real problem was simply the difficulty of carrying out such a formidable procedure. If reasonable volumes needed to be transfused, the clotting that was likely to occur meant that one had to transfuse by cannulation and connection of the artery of the donor to the vein of the recipient. This was certainly not a technique in which every physician was accomplished. It was not easy to persuade donors to have an artery cannulated and it was hard to regulate the amount transfused.
Landsteiner and the ABO Blood Group System
The necessity of transfusing blood of the same species, emphasised by Blundell, was reinforced by the observation by Crile (1869) that serum of different animals caused human red cells to clump. Landois (11) showed that animal serum caused actual lysis of human blood cells, thus explaining post transfusion haemoglobinaemia and the excretion of black urine in some early transfusions. These experiments definitively established that transfusion of animal blood to humans was absolutely contraindicated. It is thus surprising that even in the last quarter of the century some doctors, prompted by the lack of donors, were advocating transfusion of the blood of sheep.
The growth of immunology as a discipline provided clues to the reason for the incompatibility of human red cells with animal sera, since the lysis observed was considered analogous to the interaction of bacteria with antibody. Bordet found that the red cells of some species could generate antibodies in the plasma of another.
Prompted by earlier work, particularly that of Landois, Landsteiner was intrigued by the “biochemical species specificity” and considered “whether specific differentiation goes beyond the level of species, and also whether the individuals within a species show similar, though presumably slighter differences” (12). By mixing the serum and red cells of different human individuals Landsteiner established the ABO blood groups system. This obviously had great significance for the transfusion of compatible blood. Landsteiner’s work explained the failure of some early transfusions. The successes of Blundell and others were presumably due to the fortuitous use of compatible donors and recipients.
Some transfusion reactions occurred even with matched samples in the ABO system. In the 1920s Landsteiner and Levine detected other agglutinins (MN and P) in all four blood groups by injecting rabbits with human blood and showing the presence of raised antibodies to the human antigens (13).
Methods of Transfusion
Even though the matching of blood reduced transfusion reactions, at the start of the century the problem of clotting still prevented blood transfusion becoming routine. Blood clots when it comes in contact with any surface save the inner lining of blood vessels. Thus clots formed perhaps in the transfusion cannulae could enter the circulation and cause pulmonary embolism. For this reason transfusion was rarely performed, particularly when infusions of isotonic salt solutions became popular.
Carrel in 1902 (14) developed a technique in animals for joining arteries to veins, thus providing a continuous endothelial surface that enabled blood to flow from one vessel to the other without clot formation. This technique was put to practical use in a celebrated case that occurred a few years later, when Carrel was working as Research Fellow at the Rockefeller Institute. An infant of 5 days, suffering from haemorrhagic disease of the newborn, was bleeding from the nose and gut and was near death. The father, who was Professor of Surgery at Columbia University, knew of Carrel’s experiments in anastomosing blood vessels of cats and dogs, and persuaded him to anastomose his own left radial artery to the child’s right popliteal vein. The baby’s colour changed from “white to pink and finally red all over.” The infant stopped bleeding and made a complete recovery (7).
Carrel’s technique was not simple, but Crile developed an easier method using a carefully constructed ring through which a vessel could be pushed and then everted over. The other vessel could then be pushed over the ring so that the intimal surfaces of the two vessels were thus in contact. A ligature around the ring secured the anastomosis. George Crile published this work in 1907 in the Annals of Surgery, stating that the paper “is based on 225 experiments upon animals and 32 clinical cases” (15).
By the early 1900s the prevention of blood coagulation was intensively investigated. Some delay in clotting was achieved by using paraffin wax-coated vessels to collect the shed blood prior to rapid transfusion via syringe. But what was obviously required was an innocuous substance that could be added to blood to prevent clotting. Some experimenters toyed with the use of the anticoagulant substance extracted from leeches, hirudin. Lewisohn (16) tested hirudin in dogs, one of which died. Nonetheless he decided to risk the administration of a small amount of hirudin to a patient who required a transfusion following a laparotomy for carcinoma of the stomach. The patient became cyanotic, had precordial pain, imperceptible pulse and was in a precarious position for 36 hours. This effectively removed hirudin as a candidate anticoagulant, although some workers used it to rinse syringes and vessels used in transfusion in the hope that clotting might be slowed.
The breakthrough came with the discovery that citrate could prevent clotting when added immediately to freshly collected blood, and that properly citrated blood was relatively non-toxic.
The distinction of being the first to advocate the use of citrate in blood transfusion is difficult to assign. The contenders are Hustin, Agote, Weil and Lewisohn. Most accounts accept that the Belgian, Hustin (17) was the first to infuse citrated blood to a patient. However, Lewisohn probably deserves the most credit since his careful experiments established the minimum concentration necessary to prevent clotting, and the amount of citrate likely to produce a toxic effect. In his classic paper (16) Lewisohn did however acknowledge “the priority, not only for taking up this problem in a series of animal experiments, but applying it successfully in a case of human blood transfusion, belongs to Hustin, though his method, as we shall see later, limited its usefulness to small transfusions.”
Lewisohn added gradually increasing concentrations of citrate to 100 ml samples of dog’s blood. He found that whereas 0.1% citrate had no effect, 0.2% and upward prevented clotting for days. Since 1,500 ml of blood might have to be transfused, Lewisohn obtained an estimate of the likely toxic dose. He withdrew 300 ml of blood from a dog, added increasing concentrations of citrate, and re-infused the blood. He found that a total of 1.5g of citrate in the blood was fatal for a dog of 11 lbs, suggesting that 15g would be toxic for a human of 110 lbs. Even an infusion of 1,500 ml of blood containing 0.2% citrate would thus be unlikely to be toxic to humans. Next Lewisohn investigated whether the infusion of citrate might lengthen the clotting time of the recipient’s blood – certainly undesirable if the patient required a transfusion because of a haemorrhage. He found the coagulation time of blood taken from a dog was 5 minutes. He removed 300 ml of blood, added 0.2% citrate and reinfused. Samples of blood taken at 3 minute intervals after reinfusion were found, much to Lewisohn’s surprise, to have a much shortened coagulation time (10 secs).
These careful experiments, and those of Weil, who showed that citrated blood could revive guinea pigs or dogs which had been practically exsanguinated (18), paved the way for the use of citrated blood for transfusions.
As transfusion became common practice, aspects of the toxicity of citrate needed to be reassessed. During exchange transfusion in neonates, for example, it was felt that toxic levels of citrate might be reached if the transfusion was too rapid. Experiments in dogs (19) showed that infusion of citrate at a rate of 0.06 mmol/kg/min was lethal, but 0.04mmol was safe. Rates of transfusion of citrated blood were therefore kept below 0.03 mmol/kg/min citrate, or where large volumes needed to be transfused, heparinised, rather than citrated blood was used.
Similar animal experiments alerted physicians to the possible danger of transfusing large volumes of blood that had been stored for a long period. Such blood tends to have high plasma potassium levels, since this ion leaks out of red blood cells in time. Infusions in rabbits showed that whereas the citrate or potassium in transfused blood produced only mild toxicity, a combination of both killed 15 out of a group of 19 rabbits (20). Thus the toxic effects of citrate and potassium reinforced one another. Where large volumes need to be transfused therefore (as in exchange transfusion in the newborn) fresh or heparinised blood should be used. Also, where patients have already high plasma potassium, for example anuric patients or those with extensive muscle wounds, only the use of fresh citrated blood is indicated.
Storage of Blood for Transfusion
Rous and Turner in 1916 (21) noted that there had been no investigation of methods to preserve the life of red blood cells in vitro, yet their improved preservation might have practical importance, even for transfusion. If stored in the cold for too long the cells tend to lyse. After a series of simple experiments on rabbit blood, Rous and Turner found that the addition of dextrose as well as citrate protected against lysis (Rous believed that since the cells are relatively impervious to sugars they would act like a colloid and preserve the cells. Subsequent research showed however, that the dextrose has a favorable effect on red cell metabolism). Blood so treated could be stored for 15 days and was still capable of reviving severely haemorraged rabbits, which unlike control, untransfused animals, survived and showed no anaemia. Blood cells stored longer than two weeks were still able to restore the level of haemoglobin in the bled animals, but in these cases the improvement was temporary, the animals becoming progressively anaemic.
These observations on the stabilisation of red cells were put to use in World War I at a US base hospital by Oswald Robertson (22), who set up the first (albeit limited) blood bank. Robertson, during periods of relative quiet, collected blood from universal donors (selected from patients with trivial wounds or those recovered from slight wounds) direct into dextrose/citrate and placed it in an ice box. Transfusions of the stored (10-14 days) blood were given to 20 patients, mostly haemorrhage cases. The results of these transfusions were “quite as striking as those seen after transfusion with blood freshly drawn.”
The first recognised blood bank was set up in the 1930s at the Central Institute of Haematology and Blood Transfusion in Moscow. By 1937, 6,000 effective transfusions of stored blood had been made (7). The outbreak of World War II resulted in the establishment of transfusion services for the collection and storage of blood on a substantial scale.
Today, the storage and transfusion of sterile, compatible blood or blood constituents is a routine and life-saving procedure, the development of which depended upon the vision, experimentation and ingenuity of many physicians and scientists (Fig. 12.4). To suggest that blood transfusion was delayed for 200 years through dependence on animal experiments is totally incorrect. The implication of this assertion is that in 1714 transfusing coagulating blood through avian quills in non-sterile conditions could have been a routine procedure.

Fig. 12.4 Today, the storage and transfusion of sterile compatible blood or blood constituents is a routine and life saving procedure. © Science Photo Library, all rights reserved.
Such a denial of the contribution of animal experiments to human well-being stems from an unrealistic, facile conception of history.
The potent anticoagulant heparin is not widely used to prevent the coagulation of blood to be transfused. It is, however, used for venous thrombosis and to prevent blood clotting during open heart surgery and kidney dialysis.
Heparin was discovered serendipitously in the liver of dog during a search for endogenous clotting substances (23). Heparin for clinical use must be obtained from pig intestine or bovine lung, and the crude extract is standardised by measurement of its anticoagulant action on sheep plasma (BP and USP).
Opponents of animal experiments have striven vigorously to establish that the rhesus (Rh) blood group system, despite its name, owes nothing to animal experiments. In fact it is difficult to establish who first discovered the Rh antigen. The history of this interesting scientific tale is well reviewed in Mollison (7). A reasonable interpretation is that Rh antigen was discovered in Landsteiner’s laboratory in the early 1930s, by injection of rhesus monkey red cells into rabbits and guinea pigs, and adding their plasmas (containing anti-Rh antibodies) to human red cells. Landsteiner, however did not publish the work until 1940 (24).
Meanwhile Levine (a former collaborator of Landsteiner) together with Stetson, discovered an antibody (later shown to be anti-Rh) in the plasma of a woman whose child had died in utero. Since this work was published in 1939 (25), the credit for priority should go to Levine and Stetson. As far as the animal experimentation argument is concerned the question of priority is irrelevant. Levine and Stetson also described animal experiments in their paper, stating: “Agglutinins of this sort can rarely be investigated thoroughly because of their tendency to diminish in activity and eventually to disappear. Consequent attempts were made to produce a hetero-immune agglutinin of identical or similar specificity by repeated injections of sensitive blood into a series of rabbits. These experiments met with failure, since suitable absorption tests with such serums failed to reveal the presence of the desired agglutinin.” Landsteiner, using rhesus red cells as antigen, was able to raise antibodies, which enabled the distribution of Rh group system in humans to be investigated.
THE DEVELOPMENT OF BLOOD TRANSFUSION
|
The circulation of the blood (about 40 species) |
|
|
Injection into vascular system (dog) |
|
|
Transfusion of blood between dogs |
|
|
Blood transfusion revived haemorrhaged animals |
|
|
Must use blood of the same species for transfusion |
|
|
Blood transfusion saved women dying from post partum haemorrhage |
|
|
Serum of animals causes human red cells to clump |
|
|
ABO blood group system |
|
|
Technique of anastamosis of blood vessels (cat and dog) |
|
|
With pre-treatment, blood stored for two weeks (rabbits) |
|
|
Institution of blood banks for wounded soldiers |
|
|
1933 |
First large blood bank established in Moscow |
An earlier version of this chapter was published as: Animals and blood transfusion. RDS News July 1994 7-12.
References
- Sharpe R (1988) The Cruel Deception: The Use of Animals in Medical Research. London: Thorsons.
- Overell B (1993) Animal Research Takes Lives. NZ Antivivisection Society. Wellington, NZ.
- Cole FJ (1957) Harvey’s animals. J Hist Med 12 106.
- Lord Cohen (1957) ibid. 105.
- Ficarra J (1949) Evolution of Blood Transfusion, in Essays in Historical Medicine. New York: Froben Press.
- Zimmerman L & Howell K (1932) History of blood transfusion. Ann Med History 4 415.
- Mollison et al. (1987) Blood Transfusion in Clinical Medicine, 8th ed. Oxford: Blackwells.
- Coleman V (1991) Why Animal Experiments Must Stop. Green Print.
- Blundell J (1824) Some Remarks on the Operation of Transfusion, in Physiological Researches. London: E Cox & Son.
- Blundell J (1828) The Lancet ii 431.
- Landois L (1875) Die Transfusion des Blutes. Leipzig: Vogel.
- Landsteiner K (1931) Individual differences in human blood. Science 73 403.
- Landsteiner K & Levine P (1927) Further observations on individual differences of human blood. Proc Soc Exp Biol (NY) 24 941.
- Carrel A (1902) Lyon Med 98 862.
- Crile G (1907) The technique of direct transfusion of blood. Ann Surg 46 329.
- Lewisohn R (1915) Blood transfusion by the citrate method. Surg Gyn Obstet 21 37.
- Hustin A (1914) Principe d’une nouvelle methode de transfusion. J Med Brux 2 436.
- Weil R (1915) Sodium citrate in the transfusion of blood. JAMA 64 425.
- Adams WE et al. (1944) The danger and prevention of citrate intoxication in massive transfusions of whole blood. Ann Surg 120 656.
- Taylor WC et al. (1961) Experimental observations on cardiac arrhytmia during exchange transfusion in rabbits. J Pediat 58 470.
- Rous P & Turner J (1916) The preservation of living red blood cells in vitro. I Methods of preservation. J exp Med 23 219.
- Robertson O (1918) Transfusion with preserved red blood cells. BMJ i 691.
- Beck E (1984) The Treatment of Thrombosis, in Parnham M & Bruinvels J (eds.), Discoveries in Pharmacology, vol. 2. Amsterdam: Elsevier.
- Landsteiner K & Weiner A (1940) An agglutinable factor in human blood recognized by immunosera in rhesus blood. Proc Soc Exp Biol NY 43 223.
- Levine P & Stetson R (1939) An unusual case of intra-group agglutination. JAMA 113 126.







