19. Misleading Research or Misleading Statistics:
Animal Experiments and Cancer Research
Scientists supported by the Cancer Research Campaign have prepared a vaccine which it is hoped will protect against infection with Epstein-Barr virus (EBV). Apart from causing glandular fever EBV is believed to be one causative factor for certain cancers. There is firm evidence for the involvement of this virus in Burkitt’s lymphoma and in cancer of the throat and nasopharynx, and evidence for the suggested link between EBV and Hodgkin’s disease is becoming more conclusive.
Progress has recently been made in the search for a vaccine against the Epstein-Barr virus. A new vaccine has undergone Phase I clinical trials in China, where there is a high incidence of nasopharyngeal carcinoma caused by the Epstein-Barr virus (21). Progress to Phase III trials of this vaccine has been recommended by a recent meeting of cancer experts (22).
Should the vaccine prove successful, its widespread use will ultimately prevent approximately 80,000 deaths per year world-wide.
To the scientist involved in research into the prevention and cure of neoplastic disease this news will be viewed as another significant step in the steady progress made over the last 50 years in reducing mortality from cancer.
Those devoted to attacking the scientific credibility of researchers who use animals, however, look to cancer research to provide evidence to support their campaign to achieve the abolition of animal experiments.
In a recent article (1) it is claimed that despite the continued use of animals there is still an “inexorable rise in cancer mortality.” The claim is allegedly supported by selected examples of mortality figures for various cancers for 1981-85 compared to 1971-75, with the percentage change listed.
Such carefully chosen statistics provide limited information to the serious investigator. If the change in incidence is also included one can see that the increase in mortality is small in comparison with the increase in incidence (Fig. 19.1). The apparent increase in incidence is undoubtedly due to a general increase in longevity and also because the percentage of actual cancer patients surviving longer than 5 years has increased steadily in England and Wales since 1960. The more recent statistics (1988) for the USA indicate that this trend is being maintained.
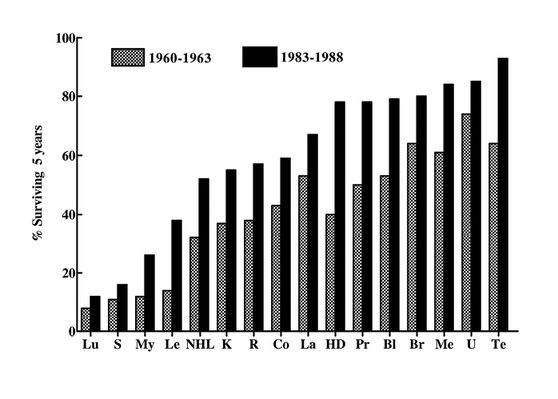
Fig. 19.1 Trends in cancer survival in the USA (male and female white), 1960-1963 and 1983-1988. Lu: lung; S: stomach; My: myeloma; Le: leukaemias; NHL: non-Hodgkin’s lymphoma; K: kidney; R: rectum; Co: colon; La: larynx; HD: Hodgkin’s disease; Pr: prostate; Bl: bladder; Br: breast; ME: melanoma; U: uterus; Te: testis.
If mortalities from cancer for different age groups are examined, changes over the last 30 years show an encouraging trend. In the age group 0-19 years mortality from cancer has dropped to under half that in 1953, in those 20-44 to 65% and in those 44-64 to 80%. Only in those aged over 65 has the mortality risen, this because of the increased longevity mentioned above. Earlier diagnosis may be a factor in the increased survival time, but undoubtedly improved treatment is significant (Fig. 19.2).
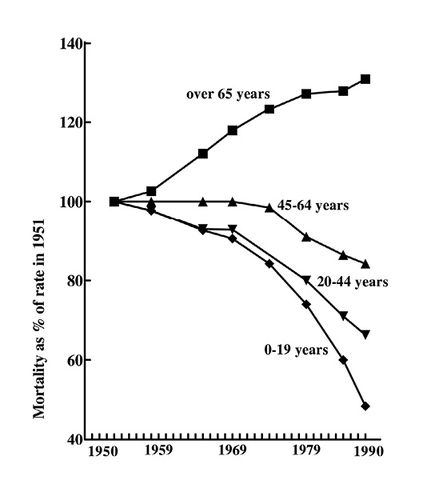
Fig. 19.2 Trends in cancer mortality in England and Wales (males), 1950-1990.
In fact, the prognosis for some cancers has improved markedly. 95% of patients with disseminated cancer of the testis are now cured,1 40-50% of patients with acute non-lymphocytic or chronic myelogenous leukaemia are also curable (2). Fortunately, the most striking progress has occurred in the treatment of childhood cancers, where 5 year survival rates have shown a striking increase over the past 30 years, with high cure rates for the childhood leukaemias and even for solid tumours (Fig. 19.3). It is perhaps significant that these statistics were not chosen for inclusion in the report referred to (1).
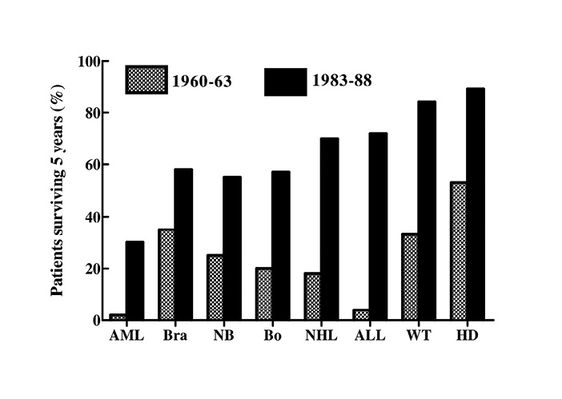
Fig. 19.3 Trends in survival of children under 15 in the USA, 1960-1963 and 1983-1988. AML: Acute myeloid leukaemia; Bra: brain; NB: neuroblastoma; Bo: bone; NHL: non-Hodgkin’s lymphoma; ALL: acute lymphocytic leukaemia; WT: Wilm’s tumour; HD: Hodgkin’s disease.
Unquestionably, the great improvements in cure rate for childhood cancer and increased life expectancy for cancer patients in general is due to the development of chemotherapy with antineoplastic agents.
Before 1940 there were no anticancer drugs. The first (1941) edition of a standard textbook on the pharmacological basis of therapeutics does not even mention cancer in the index. The eighth edition (1990) devotes 61 pages to the discussion of some 40-50 antineoplastic drugs (3).
The starting point of this dramatic change in cancer treatment was the investigation of the biological and chemical actions of the nitrogen mustards by Gilman and Philips (4). Although not published until 1946, this work was actually initiated prior to World War II due to the possible use of these vesicants in chemical warfare. For reasons of security publication was delayed.
The striking toxic action of nitrogen mustards on lymphoid tissue prompted experiments upon the effects of these chemicals on transplanted lymphosarcoma in mice (5). The suppression of tumour growth observed in this study was responsible for the first clinical investigation of a medicine to combat cancer (6).
Numerous other anticancer drugs have now been produced. Aminopterin, shown to be effective in treating various experimental neoplasms (7), actinomycin D, effective against five experimental tumours (8) and vincristine, isolated from the periwinkle (Vinca rosa). This drug was initially shown to produce bone marrow depression in animals and hence was used in an experimental leukaemia model where it was shown to be curative (9).
As knowledge of the mode of action of anticancer drugs unfolded so did the production of analogues with increased potential, either because of greater activity or diminished toxicity. This necessitated the investigation of the effectiveness of potential methods to screen compounds for activity. This was undertaken as a co-operative venture involving many national centres. Numerous in vitro systems were examined for the investigation of potential anti-tumour activity, including various mammalian cells in culture, bacteria, fungi and viruses (10). However the results indicated that no in vitro method could replace a whole animal tumour model (some drugs, cyclophosphamide for example, were only active in vivo after conversion in the liver to an active form). Furthermore, the use of no single animal tumour model could with certainty discover all the potentially useful drugs. Thus three mouse tumour models were selected for an initial screen; leukaemia L1210 (for its good predictive value for clinical activity), sarcoma 180 and adenocarcinoma 755 (these for their cytological similarity to human cancers) (10).
As more compounds were produced, tested and forwarded for clinical assessment, feedback from oncologists as to clinical efficacy enabled the experimental test systems to be reassessed and thus various other animal tumour sytems were found useful, such as the P388 mouse leukaemia, mouse B16 melanotic melanoma and the mouse Lewis lung carcinoma. The informative review by Johnson and Goldin (10) contains a table of 34 chemicals established as active against one or more human cancers. Most were effective in prolonging the life of mice with L1210 and P338 leukaemia, the majority were also active against B16 melanoma. Few clinically effective drugs failed to show an activity against at least one of the established animal tumour models.
It is of interest that one critic of the contribution of animal research towards the treatment of cancer (1) surprisingly cites the above paper as evidence of the futility of animal models, since “estrogen (sic) is effective against some cancers in women but, like prednisone fails to work in animal experiments.” To use these two drugs as evidence to diminish the value of experimental tumour systems, and to ignore the concordance of response of these experimental tumours and human cancers to the other drugs listed indicates remarkable prejudice, together with a superficial knowledge of cancer chemotherapy. Certainly, both prednisone and sex hormones were listed as inactive against the four experimental tumours examined. Prednisone and other corticosteroids seldom exert benefit in adult leukaemias and only produce cures in childhood leukaemias if given in combination with other antineoplastic agents. Oestrogen is only used for cancer of the breast (no experimental mammary cancer model was used) and then only in carefully selected patients. In fact oestrogen antagonists such as tamoxifen are more often used. (Oestrogens have of course been established as cancer causing when administered under some conditions, an action predicted by studies in the mouse by Lacassagne (11) and subsequently confirmed in other animal species).
Treatment Regimens
Animal models were not only of use for the generation of novel anticancer drugs, but were also crucial for the establishment of the principles that underpin modern cancer chemotherapy.
Using only the L1210 mouse leukaemia model, Skipper and co-workers (12) demonstrated that; 1) since a single malignant cell can divide and eventually form enough cells to kill the host, it is essential to destroy every such cell. 2) The immune system plays little or no part in the therapy of malignant disease, and 3) the destruction of cancer cells by cytotoxic drugs follows first order kinetics, i.e. a given dose of drug will kill a fixed proportion of sensitive cells regardless of the total size of the malignancy.
The consequence of these experimental studies has been to ensure total removal of malignant cells originally, by continuance of therapy after apparent remission, and later by the use of several drugs concurrently or in a logical sequence.
These studies emphasised the importance of the influence of tumour cell numbers at the start of therapy as an indicator of a favourable outcome (methotrexate, for example, was only curative in mice when treatment was started shortly after inoculation of a small number of cells). This observation did much to encourage research into methods of early diagnosis of malignant disease, since treatment at an asymptomatic stage increases the probability of a cure.
The Alternatives?
The alternative to animal experiments, assert the abolitionists, is epidemiology. To support this proposal a paper by the eminent epidemiologist Burkitt has been cited (13), in which it is argued that a “shift in emphasis towards prevention” is required. Few would dream of disagreeing that epidemiology, and ultimately prevention, is of paramount importance in the field of cancer research and indeed of most diseases. The discussion paper by Temple and Burkitt does not however support the abandonment of animal experiments. It is a plea for “simple” as against “complex” research and specifically states:
it (simple research) should use several types of investigation in parallel: (1) population comparisons, (2) prospective and case-control studies, (3) controlled clinical trials, (4) analogous studies on animals, and (5) envisaging a plausible mechanism.
It is surprising that Sharpe, in support of his condemnation of the past and future contribution of animal experimentation to research into cancer (1), should quote so extensively from a paper that specifically recommends animal experiments.
Reducing the Toxicity of Anticancer Therapy
All cancer chemotherapy is accompanied by unpleasant effects, since the drugs used are all cytotoxic, the selectivity against the cancer cells being governed by the accelerated rate of division of these cells. Research using animals has however, provided adjunctive therapy that has greatly eased the suffering of the patient.
One of the most discomforting symptoms accompanying anticancer chemotherapy or radiotherapy is nausea and vomiting. This can be combated with dramatic effectiveness with the newly developed 5-HT3 receptor antagonists. Two of these drugs, ondansetron and granisetron, are now widely used in cancer clinics to suppress completely the nausea and vomiting that accompanies treatment. The potential of these drugs was established by the demonstration that they could potently inhibit retching and vomiting induced in ferrets by cisplatin (14).
Since new blood cells are constantly being formed from rapidly dividing stem cells in the bone marrow, this organ is inevitably affected by anticancer therapy. Supportive therapy can help to alleviate this problem, for example prevention of anaemia can be achieved by the administration of factors, such as erythropoietin, that promote the formation of red blood cells. This has only been made possible by the elucidation of the circulating factors responsible for the maturation of red blood cells, starting with the significant experiments on rabbits by Carnot and Deflandre (15).
The Future
Though it is clearly justified to emphasise the progress that has been made in the treatment of cancer, it must be admitted that slow growing tumours, such as those in the lung and colon, are generally difficult to treat and are amongst the biggest killers, often because of the generation of metastases. The elucidation of the processes by which metastases develop can only be achieved by the study of tumour development and spread in laboratory animals.
A promising approach to the possibility of suppression of metastases has emerged from the observation by Folkman (16) that the antibiotic fumagillin inhibits the growth of the cells that line the inner surface of blood vessels, the endothelial cells. It is the endothelial cells that initiate the growth of new blood vessels, and tumours cannot grow above a few millimetres in size unless they can form blood vessels. A relatively non-toxic derivative of fumagillin has been shown to reduce substantially the number of metastases in mice, presumably by the inhibition of the growth of blood vessels.
Similarly one can anticipate that monoclonal antibodies (originally generated from tumour antigens) may contribute to diagnosis, imaging of tumours and to therapy, perhaps by being made to deliver a toxin or destructive radioisotope selectively to the tumour.
In some respects cancer therapy today is analogous to bacterial chemotherapy in the early 1930s. Decades of research looking for compounds that would kill the bacterial cell and leave intact the surrounding, rather similar, host tissue had produced somewhat hazardous treatments for a few conditions. But death and morbidity rates for common infections were high. Over a span of a mere seven years or so the sulphonamides and then the antibiotics had rendered infectious disease a relatively minor medical problem.
Gene therapy, the biological response modifiers and maybe a vaccine against tumour antigens (17) (in conjunction with suitable adjuvant therapy) may provide the turning point that will make the successful treatment of neoplasms routine medicine.
In recent years, the development of vaccines to prevent or treat some cancers has progressed significantly. The FDA has approved two vaccines to prevent cancer. One is a vaccine against the hepatitis B virus which can cause liver cancer (18) and the other is a vaccine against the papillomavirus, which is responsible for the majority of cervical cancer cases (19). Also, in 2010, the FDA approved the first cancer treatment vaccine for use in a particular group of men with metastatic prostate cancer. This vaccine, sipuleucel-T, is designed to stimulate an immune response to prostatic acid phosphatase, an antigen found on most prostate cancer cells (20).
Vaccines for the treatment of many forms of cancer, such as cancers of the bladder, kidney and lung, are the subject of current extensive studies.
An earlier version of this chapter was published as: Misleading research or misleading statistics – animal experiments and cancer research. RDS News April 1993 6-9.
References
- Sharpe R (1991) The War Against Cancer. Outrage. No 74 Jun/Jul.
- Chabner B, Rothenberg M (1991) Medical oncology in the 1990s. The Lancet 338, 576-77.
- Goodman L, Gilman A (1990) The Pharmacological Basis of Therapeutics. 8th Edition.
- Gilman A, Philips F (1946) The biological actions and therapeutic applications of the beta-chloroethylamines and sulphides. Science 103 409-15.
- Goodman L, Gilman A & Daugherty T (1942) unpublished observations, referred to in Goodman L et al (1946). JAMA 132 126-32.
- Gilman A (1963) The initial clinical trial of nitrogen mustard. Am J Surg 105 574-78.
- Sugiura K, Moore A, Stock C (1949) The effect of aminopterin on the growth of carcinoma, sarcoma and melanoma in animals. Cancer 2 491-502.
- Sugiura K (1960) The effect of actinomycin D on a spectrum of tumors. Ann NY Acad Sci 89 368-372.
- Johnson I, Armstrong J, Gorman M and Burnett J (1963) The Vinca alkaloids: a new class of oncolytic agents. Cancer Res 23 1390-1427.
- Johnson R, Goldin A (1975) The clinical impact of screening and other experimental tumor studies. Cancer Treatment Reviews 2 1-31.
- Lacassagne A (1936) Tumeurs malignes appareus au cours d’un traitement hormonal combine chez de souris appartenant a des lignees refractaire au cancer spontane. C R Soc Biol (Paris) 121 607-09.
- Skipper H, Schabel F (1973) Quantitative and cytokinetic studies in experimental tumor models, in Cancer Medicine eds Holland J & Frei E Lea & Febiger pp 629-50.
- Temple N, Burkitt D (1991) The war on cancer – failure of therapy and research: discussion paper. J Roy Soc Med 84 95-98.
- Bunce K. Tyers M. & Beranek P. (1991) Clinical evaluation of 5HT3 receptor antagonists as anti-emetics. TIPS 12 46-48.
- Carnot P, Deflandre C (1906) Sur l’activite hemopoietique de serum au cours de la regeneration du sang. CR Acad Sci (III) 143 384-86.
- (1991) Exploiting angiogenesis. The Lancet 337 208-09.
- Pullen L C (2011) Epstein-Barr virus vaccine may soon enter Phase 3 Trial. Medscape. Nov 07, 2011, www.medscape.com.
- Barry M, Cooper C (2007) Review of hepatitis B surface antigen-1018 ISS adjuvant-containing vaccine; safety and efficacy. Expert Opin Biol. Ther. 7, 1731-37.
- Tay EH, Garland S, Tang G et al. (2008) Clinical trial experience with prophylactic HPV 6/11/16/18 VLP vaccine in young women from the asia-pacific region. Int. J.Gynaecol. Obstet. 102 275-83.
- Kantoff P W, Higano C S, Shore N D et al. (2010) Sipuleucel-T immunotherapy for castration resistant prostate cancer. N Engl J Med 363 411-22.
- Hui E P, Taylor G S, Hui J et al. (2012) Phase I trial of recombinant modified vaccinia ankara encoding Epstein-Barr viral tumor antigens in nasopharyngeal carcinoma patients. Cancer Res 73 1676-88.
- Fauci A S, Varmus H, Nabel G and Cohen J (2011) Epstein-Barr virus vaccine may soon enter Phase III trial. Sci Transl Med 3 107fs7 (abstract).
1 “It is certainly fortunate that the platinum drugs were discovered when they were, or it (cancer of the testis) would by now be a major cause of death in young men.” Sir Richard Doll (1990) Are we winning the war against cancer? Eur J Cancer 76 500-508.







