3. Lockjaw: Prevalent but Preventable
Tetanus is one of the most dreadful diseases which has been produced in nature to torment mankind. No one who has seen a case can ever forget it. The poor victim, thrown every few minutes into the most violent spasms, bent backwards to such an extent that only his head and heels touch the bed, his teeth tightly clenched, unable to eat or drink, his face expressing the fiendish torture he is undergoing, at last is mercifully released by death.
Sir David Bruce (1920) (1)
Although tetanus was described at the time of Hippocrates, for over 2000 years there was no advance in our understanding of the disease. At the end of the nineteenth century the prevalent, rather naive view was that it was caused by “inflammation travelling up an injured nerve to the central nervous system.” (2)
The infective nature of tetanus was demonstrated by Carle and Rattone, who in 1884 took pus from a lesion of a patient with tetanus and injected it into rabbits, where it produced signs typical of the disease (3).
The Causative Organism
Tetanus frequently followed wounds where the skin had been deeply punctured, particularly if the lesions were contaminated with soil or other foreign matter such as splinters etc. Nicolaier, in 1884, established that the infective organism was present in the soil. He produced the disease in mice, rabbits and guinea pigs by inoculation of various samples of soil under the skin (4). Soil from richly manured areas, such as grazing land and the ground around stables, was particularly potent.
The identification of the bacterium responsible for tetanus was delayed because of the enormous variety of organisms present in manure-contaminated soil. Kitasato in 1889, provided definitive evidence of the identity of the causative bacterium by the use of heat (800 for one hour) to destroy non spore-bearing organisms, followed by culture in the absence of oxygen. The pure culture of “drumstick-like” anaerobic bacteria (subsequently known as Clostridium tetani) produced the typical symptoms of tetanus on inoculation into animals (5).
Tetanus Toxin
As with diphtheria, the tetanus bacillus is not invasive. It stays in the area of damage, grows and exudes a toxin which produces the symptoms of the disease. Bacteria-free filtrates of broth cultures, prepared according to the method of Kitasato, were shown to be potently toxic to animals, a few hundredths of a microlitre proving fatal to mice of 10g body weight (6).
The absorption of the toxin accounts for all the pathology of tetanus. In animals, after a latent period of 1 to 3 days, spasm occurs in the muscles at the site of the inoculation. However, should the toxin be administered intravenously, generalised muscle spasm occurs (7). This suggested that the toxin exerts its action in the central nervous system having reached there by transport along the motor nerves. This was proved by Bruschettini (8) who showed that toxin injected into muscle in rabbit could be recovered from motor nerves supplying that area. Similar animal experiments demonstrated that toxin injected into the sciatic nerve could, after a period, be recovered from the spinal cord of the animal. The fact that “local tetanus” in animals could be prevented by sclerosis or actual section of the nerve, confirmed that passage along motor nerves lying near to the focus of the tetanus infection was the means by which the toxin entered the central nervous system, where it caused its toxic actions. These experiments had profound implications for the use, and the route of administration, of the antitoxin which was developed later.
Tetanus Antitoxin
In view of the scientific climate in the late 19th century it was predictable that researchers should try to ascertain if animals could be stimulated to produce a substance that could neutralise tetanus toxin, and thus combat tetanus infection. It was perhaps also predictable that the production of a tetanus antitoxin in animals (after serial injections of gradually increasing amounts of tetanus bacillus filtrates) was demonstrated by von Behring and Kitasato, who also produced the diphtheria antitoxin (9). The antitoxin was able to protect animals against large doses of tetanus toxin or large numbers of tetanus bacilli.
Toxin was obtained by filtration of suitable cultures. The potency of the toxin was standardised by making serial dilutions and determining the minimal lethal dose (MLD) after subcutaneous injection into white mice of 20g body weight. Multiples of this MLD, perhaps rendered less noxious by treatment with terchloride of iodine, were administered to a horse in increasing doses at intervals of 8 days or more. Eventually the horse had generated sufficient antibody to withstand challenge with very high doses of tetanus toxin. The antitoxic serum was obtained by bleeding from the jugular vein and was preserved with small concentrations of phenol or tricresol. The preparation was standardised by measurement of the volume needed to protect animals (mice or guinea pigs) against lethal doses of toxin, and diluting the antitoxin solution accordingly (7).
Prophylaxis with Antitoxin
It soon became clear from experimental studies that tetanus antitoxin could only neutralise toxin that had not yet entered the motor nerves. Roux and Borrel in 1898 (10) demonstrated that rabbits with sufficient antibody in their plasma to protect them from huge doses of tetanus toxin given intravenously, readily succumbed to minute doses of toxin given by intracerebral injection. Nevertheless much animal data attested to the prophylactic value of tetanus antitoxin. The horse is particularly susceptible to tetanus infection, and tetanus following surgery or injection in this animal was commonplace. There were many reports at the turn of the century as to the prophylactic value of tetanus antiserum in the horse. A study on several hundred horses used for the production of various antisera in a large laboratory in the USA showed a 10% death-rate from tetanus despite rigid aseptic technique and disinfection. After routine prophylactic use of antitoxin before any surgical procedure, death-rate was reduced to 1% (2)
Antitoxin Prophylaxis in Humans
Although hundreds of thousands still die from tetanus infection each year, it was during war that deaths from tetanus reached epidemic proportions. Missiles such as musket balls, bullets or shrapnel, blasted through soil-contaminated clothing produce the deeply penetrating wounds in which clostridium tetani bacteria flourish. Thus military medicine provides the first clinical evidence for the efficacy of tetanus antitoxin.
According to Larrey, in Napoleon’s campaign the tetanus case mortality was 82% (cited in 1). During the American civil war case mortality was 89.3% (11, p. 203), 82% during the Franco-Prussian war of 1871 (on the German side) (1) and 90% on the British side in the Crimean war (1). The 1914-18 war was the first real testing ground for the then freely available tetanus antiserum (Fig. 3.1).
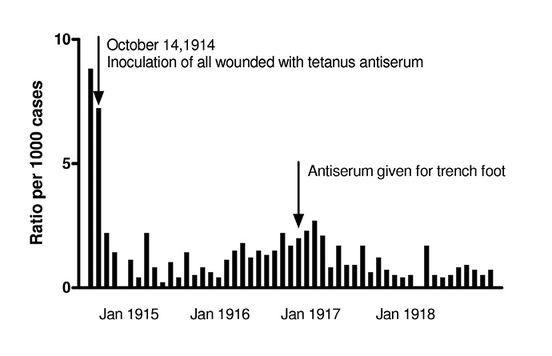
Fig. 3.1 Incidence of tetanus per 1,000 wounded in the British Army, 1914-1918.
Evidence from animal studies that the antitoxin would be effective as prophylaxis against tetanus was clear. Faced with evidence of a tetanus case mortality averaging 85%, one might have expected the British Army Medical Service in 1914 to have recommended routine administration of the antiserum to all wounded personnel. This was not the case however. For almost the first two months of the war no antiserum was used, with the result that incidence of tetanus was high. The reason for this lapse of clinical judgement was that the most recent experience of the traumas of active service was that of the Boer War. Tetanus during that campaign was rare since the veldt was clean, unlike the highly cultivated soil of Northern Europe. Although this error undoubtedly caused needless deaths during the first few weeks of the war, it also provided evidence of the clinical benefit of the antitoxin through the dramatic fall in incidence of tetanus after the belated adoption of routine anti-tetanus prophylaxis (1).
Evidence for the value of tetanus antitoxin in preventing the development of tetanus after serious wounds is undoubted, although never in fact subjected to definitive test. Without question, in medical units where all wounded were given antitoxin at the moment of entry to the field hospital, the incidence of tetanus was at most one third of that where the antitoxin was given only where the case was considered “suspicious”(12). Bazy describes a clinical situation involving 200 soldiers with similar wounds, where “on account of certain circumstances” (unspecified by the author) only 100 received antitoxin. Of these, only one developed tetanus, and that very soon after the injection, suggesting that the disease had already taken hold. Of the 100 left unprotected, 18 developed tetanus. Thus the “certain circumstances” that obtained in Bazy’s study, whilst unfortunate for 18 subjects, provided something akin to a partially controlled clinical trial to demonstrate the efficacy of tetanus antitoxin (12).
Therapy with Antitoxin
The value of antitoxin to treat tetanus once the central nervous effects of the disease are manifest is less easy to establish. The heterogeneity amongst patients with tetanus and the ethical problem of withholding a potentially life-saving treatment mean that a properly controlled trial is hard to conduct. One such, assessed by sequential analysis, did show a significant benefit from administration of 200,000 units of antitoxin (13). The small number of patients in this trial (38 pairs) has meant that the results are not accepted everywhere.
In view of the knowledge of the nature of the progress of infection by tetanus (derived solely from animal experiments) one might have supposed that injection of antitoxin direct into the nervous system would be attempted, since this is where the toxin is exerting its pathological effects. Indeed, in a particularly rigorous experiment, Sherrington (14) showed that intrathecal injection of antiserum to rhesus monkeys could reduce mortality to a substantial dose of toxin from 100% to 48%, even if the injection of the antidote was delayed until overt signs of tetanus were apparent.
It appears, however, that physicians were loath to administer a foreign protein (i.e. equine antitoxin) into the human central nervous system. Some trials carried out in India (15) demonstrated that the equine antitoxin given intrathecally was beneficial if administered less than 24 hours after start of symptoms, and a report from Irwin Hospital showed that intrathecal injection of human tetanus immune globulin reduced mortality from “mild” tetanus from 21% to 2% (15).
It is clear that tetanus antitoxin is prophylactic if administered in repeated doses to patients at risk of infection. However, once the disease has taken hold, antitoxin will be of use only to neutralize toxin that has not yet entered the nervous system, or if given intrathecally, to negate the effect of toxin that is in the nervous system but not yet fixed to the tissues. Complete control of the disease could only be achieved by effective immunisation programmes.
Immunisation against Tetanus
Effective immunisation against tetanus became possible when Descombey (16) demonstrated that tetanus toxin was rendered innocuous after prolonged incubation with formaldehyde (thus forming “toxoid”). Bergey and Etris (17) showed that three doses of toxoid, given over a period, could protect guinea pigs against injection of several thousand times a lethal dose of tetanus toxin. Since the 1930s toxoid, or toxoid precipitated with alum or other agents, has been widely used in humans.
Preparation of Toxoid
Tetanus vaccine is prepared as prescribed by the World Health Organisation (18). Tetanus toxin is obtained from a liquid medium in which a high-yielding strain of clostridium tetani has been cultured. The toxin is converted to the non-toxic “toxoid” by treatment with a dilute solution of formaldehyde for days or weeks. During this time the product is continually tested for toxicity in animals. The complete conversion of toxin to toxoid is established by injection of suitably diluted samples into 5 guinea pigs. The sample of toxoid passes this formal “specific toxicity test” if no guinea pig shows sign of paralysis or any other sign of tetanus within 21 days. Potency tests (protection of guinea pigs against challenge with a subcutaneous injection) and a test for innocuity (intraperitoneal injection to mice and guinea pigs) are also stipulated by the WHO monograph and national pharmacopoeias.
The Success of Tetanus Immunisation
Routine vaccination against tetanus has been the norm in developed countries for three or four decades. Effectiveness of the procedure is generally assessed by the presence of high antibody titres in the blood. Due to the consequent rarity of the condition in the developed world, as with antitoxin, one has to look to war to provide evidence of the efficacy of vaccination.
Unfortunately, during war reliable data is not always accurately recorded. Nevertheless a good account of the incidence of tetanus in the US Army is reported by Long and Sartwell (19). All US troops were immunised with tetanus toxoid from 1941. During World War I, when troops were non-immunised, out of 523,158 wounded 70 developed tetanus. During World War II 2,734,819 soldiers were wounded, yet only 12 cases of tetanus occurred, and six of these occurred in subjects who were not in fact immunised, for a variety of administrative reasons. In the Pacific theatre incidence of tetanus was high amongst non-immunised troops and civilians. In a group of 550 wounded Japanese prisoners of war in the Marshall Islands, 26 developed tetanus, and tetanus caused approximately 400 deaths of civilians injured in the war zones of Manilla and Saipan. Other comparative data attest to the protective value of vaccination against tetanus (20, 21)
Neonatal Tetanus
Current estimates of mortality from tetanus are 800,000 deaths per year from neonatal tetanus, and 120,000 to 300,000 deaths from non-neonatal tetanus (22). The cause of neonatal tetanus is generally due to infection of the umbilical scar caused by poor obstetric technique. Tetanus accounts for approximately 50% of neonatal deaths and 25% of infant deaths (22). As noted by Habig and Tankersley, “this high incidence is additionally tragic because the disease is largely preventable by appropriate immunisation.” (23)
Conclusive proof of the value of tetanus toxoid in the prevention of tetanus is provided by a double-blind controlled field trial for the prevention of tetanus neonatorum in Colombia (South America), where in 1966 the estimated death rate from the condition was 11.6 per 100 births. Over 2.5 thousand women were initially involved in the trial. The control group received injection of a polyvalent influenza-virus vaccine, the test group aluminium-phosphate-adsorbed tetanus toxoid. In children born to women in the control group, incidence of neonatal tetanus was 7.8 per 100 births (27/347). None of the 341 children born to mothers previously given 2 or 3 doses of tetanus toxoid developed the disease. Non-tetanus mortality in the two groups was similar (5.5 and 4.1% respectively) (24, Table 1).
THE USE OF TETANUS TOXOID FOR THE PREVENTION OF NEONATAL TETANUS. Report of a double-blind controlled field trial covering 1618 women in a rural area of Colombia. From Newell, Duenas Lehmann, Leblanc & Garces Osorio (1966) Bulletin of the WHO 35 863-71.
|
Control |
Vaccinated |
|
|
Total births |
347 |
341 |
|
Tetanus mortality (%) |
27 (7.8) |
0 (0) |
|
Non-tetanus mortality (%) |
19 (5.5) |
14 (4.1) |
Management of Tetanus
Knowledge obtained through animal experimentation has not only contributed to development of tetanus antitoxin and toxoid, but also to other modes of treatment. Antibiotics (penicillin and metronidazole) may be given to prevent the growth of clostridium tetani and so prevent further accumulation of toxin. Should the disease have taken hold and convulsions started, neuromuscular blocking drugs must be used to prevent spasm, and breathing maintained by positive pressure ventilation (25). The development of both antibiotics and neuromuscular blocking drugs depended totally on animal experimentation.
The Action of Tetanus Toxin
Many potent toxins of plant and animal origin have proved to be valuable tools to elucidate cell and organ function. Botulinus toxin and black widow spider venom are two notable examples. Tetanus toxin is no exception. It has been shown to prevent the release of chemical transmitters from nerves by a highly selective action on a protein present in the outer membrane of the vesicle that encloses the stores of transmitter in nerve endings. Presumably the toxin has a particular affinity for those nerves in the spinal cord that release inhibitory transmitters. Disruption of activity of these nerves would cause overactivity of motor nerves and generalised spasm. Recent research in rat tissue (26) shows that tetanus toxin is a zinc endopeptidase enzyme that selectively destroys the membrane protein, synaptobrevin. Use of the toxin will help to unravel the various functions of the different constituents of the vesicular membrane. With such tools we shall eventually elucidate the esoteric, intracellular processes controlling the storage and release of chemical messengers from nerve cells. We may also, incidentally, expose new methods of treatments for tetanus, for example the use of zinc endopeptidase inhibitors such as the antihypertensive drug, captopril.
Tetanus toxin was isolated 30 years before chemical transmission of nerve action was even raised as a possibility. The fact that some 60 years on, we are anticipating the use of this toxin in the elucidation of intricate molecular processes governing release of neurotransmitters from individual nerve cells is an example of the remarkable exponential nature of the progress of research.
Tetanus Today
Tetanus still occurs, even in developed countries. Typically it strikes females over 50 years of age, frequently infected after a minor cut or abrasion sustained while gardening. This susceptibility is due to the fact that routine tetanus immunisation of infants began only in the 1950s. Thus, those too old to have benefited from this, and who did not receive the vaccine as a member of the armed forces, would be vulnerable. Three recent cases have been reported in patients over 60 (27). This argues for vaccination for those unprotected and who are at risk by virtue of their job or hobby.
Tetanus vaccination appears to have been ignored by the animal rights lobby. No attack upon its usefulness can be found after a brief examination of recent antivivisection literature. This is probably due to its undoubted efficacy, its remarkable safety (side effects are virtually non-existent) and to its widespread use to protect animals against the disease. The Index of Veterinary Specialities lists 20 vaccine preparations containing tetanus toxoid, alone or in combination with vaccines against other animal diseases (28).
An earlier version of this chapter was published as: Lockjaw: prevalent but preventable. RDS News April 1994 6-9.
References
- Bruce D (1920) The Prevention of Tetanus during the Great War by the Use of Antitetanic Serum. London: Pulman & Sons.
- Paget S (1906) Experiments on Animals. 3rd ed London: James Nesbit & Co.
- Carle and Rattone (1884) Giornale dell’Accademia di Medicina di Torino. 3rd series 32 174.
- Nicolaier A (1884) Ueber infectiösen Tetanus. Deutsch Med Woch 10 842-44. http://dx.doi.org/10.1055/s-0028-1143432
- Kitasato S (1889) Ueber den Tetanusbacillus. Zeit fur Hyg 7 225-34. http://dx.doi.org/10.1007/BF02188336
- Brieger and Cohn, cited in Zinsser H (1931) Textbook of Bacteriology. 6th edition New York: Appleton.
- Zinsser (as above).
- Bruschettini A (1890) Diffusione negli organi del veleno tetano. Riforma Medica 6 1346.
- Von Behring E & Kitasato S (1890) The mechanism of diphtheria and tetanus immunity in animals. Deutsch Med Woch 16 1113.
- Roux E & Borrel A (1898) Tétanos cérébral et immunité contre le tétanos. Annales de l’Institut Pasteur 12 225.
- Keen W (1914) Animal Experimentation and Medical Progress. Boston: Houghton Mifflin.
- Bazy M (1914) Comp Rendus Soc Biol 159 794.
- Brown A et al. (1960) Value of a large dose of antitoxin in clinical tetanus. The Lancet ii 227.
- Sherrington CS (1917) Observations with antithetanus serum in the monkey. The Lancet ii 964-66.
- Annotation. The Lancet ii 464, 1980.
- Descombey P (1924) L’anatoxine tétanique. Comp Rendus Soc Biol 91 239.
- Bergey DH & Etris S (1933) Tetanus toxoid in prophylaxis against tetanus. J Infect Dis 53 331. http://dx.doi.org/10.1093/infdis/53.3.331
- (1979) WHO expert committee on biological standardization. Thirtieth report, technical report series 638 World Health Organization, Geneva. http://dx.doi.org/10.1016/0016-6480(80)90066-0
- Long A & Sartwell P (1947) Tetanus in the United States Army in World War II. Bull US Army Med Dept 7 371.
- Christenson B & Bottiger M (1987) Epidemiology and immunity to tetanus in Sweden. Scand J Infect Dis 19 429.
- Simonsen O et al. (1987) Epidemiology of tetanus in Denmark 1920-1982. Ibid. 19 437.
- Cryz S (1991) Vaccines and Immunotherapy. New York: Pergamon.
- Habig W & Tankersley D (1991) (in Cryz above).
- Newell K et al. (1966) The use of toxoid for the prevention of tetanus neonatorum. Final report of a double-blind controlled field trial. Bull WHO 35 863.
- Smith J & Collee J (1990) Tetanus, in Topley & Wilson’s Principles of Bacteriology, Virology & Immunity, vol. 3. London: Edward Arnold.
- Schiavo G et al. (1992) Tetanus and botulinum-B neurotoxins block neurotransmitter release by proteolytic cleavage of synaptobrevin. Nature 359 832.
- The Independent 10 August 1993.
(1991) Index of Veterinary Specialities 31 no 1.







