4. Pertussis Vaccine, Unfairly Maligned – At What Cost?
Whooping cough prophylaxis, as pertussis vaccine, is routinely administered to infants together with diphtheria and tetanus as the DPT combined immunisation.
After a short catarrhal phase, whooping cough begins with a dry nocturnal cough. This progresses to prolonged bouts of coughing which often end with a sharp intake of breath. The coughing fits may induce vomiting and, in severe cases, convulsions. Frank damage to the nervous system may occur, this is due to haemorrhage and lack of oxygen in the brain caused by the raised venous pressure that occurs during the paroxysms.
The symptoms persist for weeks and may be complicated by pneumonia, bronchitis and collapse of the lung. Admission to intensive care is frequently required. The patient may be left in a debilitated condition with an increased susceptibility to pulmonary infections. Whooping cough is without doubt an extremely distressing disease for both the patients and carers.
Prior to vaccination, mortality to whooping cough was the highest for any childhood disease. In Europe and the USA between 1900 and the 1920s the annual death rate was between 10 and 11 per 100,000 (mostly children under 3 years). In 1931, a standard textbook of bacteriology stated: “[whooping cough] may be looked upon as one of the major causes of death in civilised countries.” At this time it was responsible for 1.3% of all deaths in England and Wales (1). Today, whooping cough is still a significant cause of child mortality in developing countries. In 1991 it was estimated that, world-wide, pertussis caused the deaths of approximately 340,000 children each year (25).
The causative organism of whooping cough was described in 1900 when Bordet and Gengou observed small, ovoid bodies in the sputum of a child with the disease. Cultivation of the bacillus (Bordetella pertussis) was not achieved until 1906 when the same workers managed to grow the organism on a composite medium of glycerin extract of potato, agar and defibrinated rabbit blood (2).
Toxins obtained from these cultures were lethal to rabbits after intravenous injection. Subcutaneous injection produced a non-suppurating local necrosis (1). The pathological effects in humans are presumably due to local necrosis caused by toxins released by the bacilli growing on the ciliated cells lining the bronchi and trachea. A thick, ropy exudate is formed and the attempted ejection of this, together with a systemic effect of pertussis toxin, is the cause of the paroxysmal cough.
The Vaccine
It was natural that researchers should attempt to develop a vaccine that would protect against such a distressing disease with a high child mortality. Clinical trials began in the 1920s to determine the protective effect conferred by extracts of killed Bordetella pertussis. The vaccines were generally considered to confer some degree of protection, although the studies were not always well-controlled (3).
To assess the potential of the various vaccines against whooping cough, a series of rigorous trials under the control of the Medical Research Council began in England after World War II and were completed in 1959. This whole investigation was of impeccable quality and reflected great credit on the relevant committee of the MRC.
It soon became apparent that the candidate vaccines varied in ability to protect against the disease. It was thus decided to assess the correlation between the efficacy of the vaccines in children and the potency in the “mouse protection test.” In this test, groups of 15 mice were injected with graded doses of vaccine, and 10-14 days later challenged intracerebrally with a virulent strain of the bacillus. From the proportion of mice dying in each group an estimate of the efficacy of the vaccine could be obtained. In a study of 25 vaccines there was a high degree of correlation between the activity in the mouse test and the effectiveness of the vaccine in the field (4). These meticulous observations thus provided a quick method of assessment of new vaccine preparations, and the final report of the MRC trials recommended (5): “Only those pertussis vaccines which have been shown, by the intracerebral mouse protection test, to have adequate potency in relation to the British Standard Pertussis Vaccine should be used in whooping cough prophylaxis.”
The MRC trials, double-blind and randomised as they were, showed definitively that vaccination against pertussis was effective. Immunisation of infants was therefore officially recommended in 1957, although immunisation had already spread throughout some areas of England and Wales during the 1940s and early 1950s as local health authorities gradually adopted a pertussis immunisation programme and replaced diphtheria/tetanus vaccine with diphtheria/tetanus/pertussis vaccine in their routine procedures (6) (Fig. 4.1).
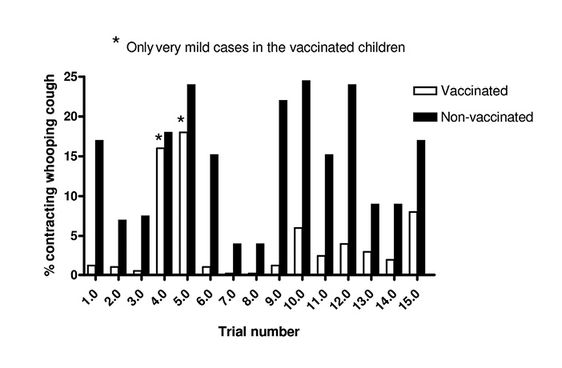
Fig. 4.1 Studies on pertussis immunisation, 1937-1942. 1.0 Saur 1938; 2.0 Park 1937; 3.0 Kendrick Eldering 1939; 4.0 Doull-Shipley 1939; 5.0 Kramer 1938; 6.0 Silverthorne 1939; 7.0 Siegel 1938; 8.0 Siegel 1938; 9.0 Singer 1940; 10.0 Howell 1938; 11.0 Kendrick Eldering 1939; 12.0 Kositza 1940; 13.0 Lapin et al 1939; 14.0 Lapin et al 1939; 15.0 Perkins 1942.1
The beneficial effect of routine vaccination against whooping cough is well illustrated by the drop in incidence in England and Wales as the vaccination acceptance rate rose to around 80% (Fig. 4.2).
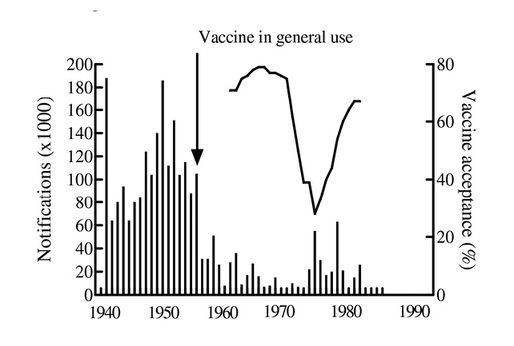
Caption: Fig. 4.2 Whooping cough notifications in England and Wales, 1940-1990.
Clear confirmation that the drop in incidence was not fortuitous coincidence is provided by the upturn in incidence as the vaccine acceptance rate unfortunately dropped between 1975 and 1983 (7). Elsewhere, in countries such as Fiji, where the vaccination programme was not interrupted, the disease was practically eliminated (8) (Fig. 4.3).
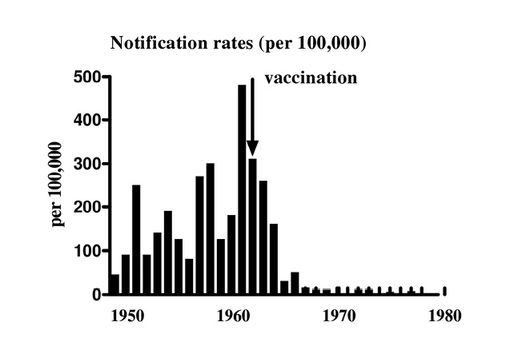
Fig. 4.3 Whooping cough in Fiji, 1950-1980.
Questions of Safety
Undoubtedly, the killed, whole cell pertussis vaccine is highly reactogenic. Redness and soreness at the site of injection and fever are common. Incidence of serious reactions such as persistent screaming, convulsions or shock accompanied by collapse have been variously estimated as 1 in 5000 to 1 in 50,000 injections (9), although these studies were never controlled and thus neglected the natural incidence of these events. For example, a first convulsion may occur naturally in approximately 1 child in 400 aged between 3-9 months. Thus one child in 10,000 in that age group, given 3 doses of vaccine might by chance have a first convulsion within 3 days of an injection (7).
The great contention that has dogged discussion of the safety of pertussis vaccine for the last two decades is whether it occasionally produces irreversible brain damage. The history of the rise and fall of this canard should be mandatory study for both medical students and science writers.
There were sporadic reports of an alleged association between pertussis vaccination and serious brain damage but these were paid little heed since control studies were never performed and most informed authorities did not assume a causal relationship (9). The discussion was kept within the medical establishment and thus did not affect the acceptance rate of pertussis vaccination by the public.
A crucial event was the publication in 1974 of a paper that stated that over an 11 year period, 36 children were admitted to a large London hospital with acute neurological illness that had started within 28 days of an injection of pertussis vaccine (10). The authors made no claim that the vaccination caused the illness and in some of the cases there were probable diagnoses that excluded an assumption of a causal relationship. This report might well have remained accessible only to those capable of assessing the significance of these observations in conjunction with other valuable epidemiological studies. Unfortunately the mass media gave them much publicity with the result that the general public and some general practitioners (but not consultant paediatricians) were reluctant to accept pertussis immunisation.
Between 1977 and 1983 the question of whether or not pertussis vaccine caused irreversible brain damage continued to be argued. On one side allegations of an incidence of 1 in 60,000 to 1 in 10,000 were quoted (11); on the other the possibility was described as “not proven” or very rare (1 in 135,000 to 1 in 300,000) (12, 24). The problem was the temptation (to some) to accept that any neurological defect that developed after vaccination was necessarily caused by it. Natural incidences of neurological problems in infants were ignored. The diagnostic problems of assigning a severe encephalopathy to vaccination were well illustrated by the clinical account of 3 children who developed mental handicap, symptoms of which appeared 6 hours, 2 days and 1 week respectively after “the whooping cough jab” (12). These children might well have remained designated as “vaccine damaged” but for follow up studies one and a half to four years later, which showed two of the children had suffered prenatal damage and the third had X-linked Menkes disease inherited from his maternal grandmother. In the same paper the pertinent point was made that if, as some suggested, 1:20,000 children vaccinated with pertussis suffered brain damage, then there should be enough affected children for each specialist in paediatric neurology to have an appreciable case load of children with a characteristic disease indubitably associated with vaccination. This was not the case.
The relative danger of forgoing vaccination for a perceived risk of, say a 1 in 135,000 chance of serious neurological damage, were well illustrated by Grist in 1977 (13). He claimed that 135,000 cases of whooping cough “would entail 30 cases of encephalitis..., many cases of convulsions, 170 deaths and 13,500 illnesses requiring hospital admission; many children would have pneumonia, bronchitis, and lung damage, and most of them would experience distressing symptoms for many weeks.”
The safety of pertussis vaccine has now been established following the comprehensive survey by Pollock and Morris (14) which found no evidence that DPT caused major neurological damage. This has been confirmed by two recent large scale studies from Britain (15) and the USA (16). The first concludes “(the) study has clearly shown that illnesses leading to death or brain damage after diphtheria, tetanus, and pertussis vaccine, if they occur at all, are extremely rare.” The second, which involved 218,000 children, “did not find any statistically significant increased risk of onset of serious acute neurological illness in the 7 days after DTP vaccine exposure for young children.”
The reassurance provided by these thorough studies comes years too late for the 102,000 children who became seriously ill (32 died) in the epidemic of 1977-79, which coincided with the dramatic decline in vaccine acceptance due to misplaced publicity of brain damage incorrectly attributed to the vaccine. As others have noted with some cynicism (17, 18), the good news of the establishment of the safety of pertussis vaccine has apparently gone unnoticed by the media. Fortunately, the information was actively promoted by the Department of Health with the result that confidence in the vaccine returned and the acceptance rate has risen to 92%.
The heated epidemiological arguments over the safety of pertussis that waged for 10-15 years provided an opportunity for the promulgation of persuasive propaganda by those opposed to animal experiments. Sharpe, for example (21), quotes only data from Stewart, who was virtually the sole advocate of the rejection of the vaccine on grounds of safety. This view was emphatically but courteously attacked in the medical journals by both neurologists and paediatricians (12, 13, 19, 20). This contention was quite overt and was not only carried out in the correspondence columns of the British Medical Journal and the Lancet, but also featured in editorials and leading articles. These discussions are ignored by Sharpe, as is the substantial study by Pollock and Morris (14) (which absolves the vaccine) and the scholarly and well-balanced review by Miller, Alderslade and Ross (9), both of which appeared 5 years before the publication of Sharpe’s attack on animal experimentation, The Cruel Deception. The misrepresentation that results from selection of evidence solely supporting one’s prejudice is in itself a deception.
Sharpe also questions the benefit produced by pertussis vaccination. He presents a graph (with no scale or actual figures) which shows a steep fall in mortality due to whooping cough from 1860 onwards (23). He thus claims that the disease had ceased to be a problem before the advent of antibiotics or vaccination. Mortality certainly had fallen due to great improvements in the techniques applied to the care of infants with respiratory disease. Application of specialist nursing, techniques of aspiration, the availability of intensive care units, oxygen etc., all contributed, as did the availability of antibiotics to combat secondary infections. Nonetheless to suggest that the disease was of little significance is a travesty.
The incidence of the disease did not diminish until vaccination. Even if mortality was reduced, whooping cough was a severe and dangerous disease in a substantial proportion of affected children, as the epidemic of 1977-79 showed. The problem was well illustrated by the Royal College of General Practitioners special report on the nature of this epidemic in a large population of West Glamorgan (22). 2,295 cases were reported, two children died of apparent cot death, 64 unvaccinated children had to be admitted to hospital, 24 needed aspiration, 25 intensive care, 10 required oxygen. Two had encephalitis (one died), 14 had convulsions, 224 had acute bronchitis, 8 had collapsed lung, 17 developed asthma; there were 26 cases of apnoea. Whooping was present in 45% of cases, the paroxysms were followed by vomiting in 66%. Severe cyanosis after paroxysms occurred in 430 cases. The avoidance of morbidity and mortality of this magnitude would appear a sound justification for the adoption of pertussis immunisation.
The Future
The anxiety over the possible severe adverse effects of the whole cell pertussis vaccine, needless as it was, nevertheless reduced public confidence in the vaccination programme. It is thus important to restore this confidence by the development of a vaccine which contains the antigenic fractions, and no active pertussis toxin or extraneous matter that is likely to cause the local reactions and fever.
A number of candidate vaccines have been produced based on antigenic fractions of precipitates of centrifuged suspensions of bacteria (23). The possible value of these vaccines can be rapidly assessed by their ability to protect mice against intranasal challenge with B pertussis or direct challenge with pertussis toxin. The possibility of reversion to frank toxicity, which has been observed with some potential pertussis vaccines, can be excluded by two simple tests in mice; the histamine sensitisation test or the measurement of leukocytosis-promoting activity (23). An in vitro test is now available for pertussis toxin based on the morphological changes it produces in Chinese hamster ovary cells.
An earlier version of this chapter was published as: Pertussis vaccine, unfairly maligned – at what cost? RDS News April 1994 10-13.
References
- Zinssner H (1931) Textbook of Bacteriology. 6th ed. New York: Appleton.
- Bordet & Gengou (1906) Le microbe de la coqueluche. Ann de l’Inst Pasteur 20 731.
- Lapin J (1943) Whooping Cough. Springfield: C C Thomas.
- Medical Research Council (1956) Vaccination against whooping cough. Relation between protection in children and results of laboratory tests. Br Med J ii, 454
- Medical Research Council (1959) Vaccination against whooping cough. Final report. Br Med J i, 454-62. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2034830/
- Griffith A (1982) ABC of 1 to 7: whooping cough (Letter). Br Med J 284 1263-64.
- Parker M & Collier L (1990) Topley & Wilson’s Principles of Bacteriology, Virology and Immunity: Systematic Bacteriology. 8th Edition Volume 3. Hodder Arnold.
- Pollard R (1983) Whooping cough in Fiji. The Lancet i 1381.
- Miller D, Alderslade R & Ross E (1982) Whooping cough and whooping cough vaccine. The risks and benefits debate. Epidemiol Rev 4 1.
- Kulenkampff M, Schwartzman J & Wilson J (1974) Neurological complications of pertussis inoculation. Arch Dis Child 49 46.
- Stewart G (1977) Vaccination against Whooping-cough. Efficacy Versus Risks. The Lancet i 234.
- Stephenson J (1977) Vaccination against whooping-cough. The Lancet i 357.
- Grist G (1977) Vaccination against whooping-cough. The Lancet i 358.
- Pollock T and Morris J (1983) A 7-year Survey of disorders attributed to vaccination in north west Thames Region. The Lancet i 753.
- Miller D et al. (1993) Pertussis immunisation and serious neurological illness in children. Br Med J 307 1171.
- Gale J et al. (1994) Risk of Serious Acute Neurological Illness after Immunisation with DPT Vaccine. JAMA 271 1.
- Paton W (1993) Man and Mouse: Animals in Medical Research. Oxford: Oxford University Press.
- Minerva Med (1983) Views. Br Med J 286 1288, http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1547286/?page=1
- Miller D and Ross E (1982) ABC of 1 to 7: Whooping cough. Br Med J 284 1874.
- Preston N (1982) Toxicity of pertussis vaccine. Br Med J 284 1817.
- Sharpe R (1988) The Cruel Deception: The Use of Animals in Medical Research. London: Thorsons.
- Royal College of General Practitioners (1981) Report from the Swansea Research Unit. Effect of a low pertussis vaccination uptake on a large community. Br Med J 282 23. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1503761/
- Cryz S (1991) Vaccines and Immunotherapy. New York: Pergamon.
- Valman H (1982) Whooping cough. Br Med J 284 886.
- Galazka A (1992) Control of pertussis in the world. Wld Hlth Statist Quart 45 886.
1 Data taken from J H Lapin (1943) Whooping Cough. London: CC Thomas, from which complete references may be obtained.







