9. The Contribution of Animal Experiments to Kidney Transplantation
Haemodialysis is life-saving and curative in acute renal failure. By reversing the build-up of metabolic products normally excreted by a functioning kidney, dialysis enables the temporarily affected kidneys to heal and resume normal function. In chronic renal failure however, the burden of regular dialysis is necessary unless a healthy kidney from a donor can be grafted.
Chronic renal failure (CRF) due to glomerulonephritis, pyelonephritis or polycystic kidney disease is quite common, particularly in young adults. Depending on the composition of the population, between 50 and 100 persons per million will develop chronic renal failure each year (1).
In the absence of treatment, the deterioration in kidney function results in anaemia and weakness. Since water cannot be excreted, fluid accumulates in the tissues and consequent lung oedema causes breathing difficulties and strain on the heart, an organ probably already compromised by high blood pressure, which is a common feature of kidney failure. The build-up of waste products results in inflammation of various tissues leading to pericarditis, colitis, diarrhoea, gastritis and persistent vomiting. Peripheral neuritis can result in paralysis (2).
Experiments in Transplantation
The miserable clinical picture of the patient, often young, sinking inexorably into coma, and from coma to death was a potent stimulus to research. The possibility of treating CRF by grafting a healthy kidney was a natural experiment to attempt. However, the transplantation of a large organ such as the kidney, which receives its blood supply via a single, large artery, requires the connection (or anastomosis) of the renal artery and vein of the donor kidney with a suitable artery and vein in the patient. This was no simple task since the arterial anastomosis had to be secure enough to withstand systolic blood pressure.
This formidable problem was resolved by the French surgeon Alexis Carrel (Fig. 9.1), who in 1902 described a method for suturing together the cut ends of blood vessels (3).

Fig. 9.1 Alexis Carrel, 1912 Nobel Laureate in Physiology or Medicine.
Wellcome Library, London, CC BY.
Carrel initially fixed three sutures equidistant around the end of each vessel and applied tension, thus forming a triangle. Corresponding sides of the triangulated vessels were then sewn together using extremely fine needles and the thinnest linen thread as used by the lace makers of Valenciennes (Fig. 9. 2). Carrel performed many anastomoses, of various types, in cats and dogs. These healed and functioned well for many months (4). It is noteworthy that Carrel emphasised the importance of protecting the endothelial cell layer during the procedure. A fact he found by experiment, but obvious today in view of the many important factors known to be synthesised and released by these cells.
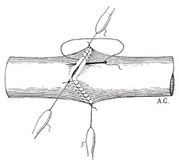
Fig. 9.2 Carrel’s vascular anastamosis. From A. Carrel (1902), ‘La Technique operatoire des anastomoses vasculaire et la transplantation des visceres’, Medecine de Lyon, 98, 859.
Carrel used his suturing technique to experiment with kidney transplantation in dog and cat. His anastomoses enabled a kidney, removed and transplanted to a different site in the same animal (an autograft), to function adequately provided that the organ was not deprived of a blood supply for too long. However, a kidney grafted from another animal of the same species (a homograft or allograft) functioned for only a few days. After that it would become oedematous and cease to work. After many experiments, Carrel became convinced that it was not the surgery that caused the problem, since autografts always functioned reasonably well. He concluded that the changes that occurred in the kidney transplanted from a different animal of the same species were due to “biological factors” from the host. Thus the phenomenon of rejection was exposed (5).
Rejection
Carrel was the first scientist in America to be awarded the Nobel Prize. This was in 1912 for work on “vasculature suture and transplantation of organs.” He returned to France at the outbreak of World War I. Although he went back to the USA in 1919, he did not return to the problem of kidney transplantation, but worked on the in vitro cultivation of organs and tissues.
The inevitability of the rejection of homografted kidneys was confirmed in 1923 by Williamson, a surgeon working at the Mayo Clinic Foundation. Williamson’s experiments in dogs were similar to those of Carrel. They were slightly more sophisticated, however, in that he analysed the urine produced by the kidney homografts and autografts, and noted that there was very little difference until rejection began in the homograft. Williamson also examined microscopic sections of the homografted kidneys and was thus the first to describe the histological picture of the kidney during rejection (6).
The seemingly insuperable problem of the refusal by a host to tolerate the presence of an organ from another animal from the same species caused a hiatus in transplantation research. It was not for another 20 years that the dreary clinical picture, and the frustration of the physician attempting to treat CRF, again stimulated research into the possibility of kidney transplantation.
There were some sporadic attempts to transplant kidneys into seriously ill patients. The thigh was usually chosen as the transplant site. This was anatomically inconvenient, but the ease of access meant that the kidney could be easily examined and removed without major surgery. As might have been predicted, long-term survival was not attained.1
Perfection of the Surgery
Although, in experimental animal studies, autografted kidneys worked adequately, they did not function in a completely normal way. This was probably due to the absence of the nerve supply, lymphatic drainage and various other factors. The surgeon Joseph Murray joined the Brigham Hospital in Boston in 1951, and initially worked in the laboratory to determine the optimum surgical technique for kidney transplantation. Murray demonstrated in dog that the best place for the graft was in the abdomen, behind the peritoneal membrane and connected to the blood vessels of the pelvis. The ureter could then be sutured to the bladder, instead of to the skin, as in the early transplants (the latter technique frequently resulted in infection). By 1954 Murray had several dogs living for over 2 years on a life-sustaining renal autograft (7).
Armed with a perfected technique, and impressed with the knowledge that a kidney allograft in a dog with both kidneys removed would excrete urine and support life until rejection, the surgeons at the Brigham began in earnest to attempt transplantation in patients with CRF. Without any attempt to tackle the problem of rejection these were obviously not successful. Nevertheless important lessons were learned. As in the animal studies, the transplanted kidney could restore the uraemic patient to normal and Murray’s placement of the kidney in the abdomen, and the anastomosis with pelvic vessels perfected in dog, worked well in patients. The necessity of sometimes removing the diseased kidneys to prevent hypertension was established, and the pathological changes occurring in the kidney during rejection were observed and shown to be similar to those in the animal studies.
The Mechanism of Rejection: An Immunological Phenomenon
Clues to the mechanism of the rejection of homografted tissue had actually appeared in the literature long before surgeons were seriously considering kidney grafting for CRF.
In 1916 Little and Tyzzer (8) had shown that tumours that arose spontaneously in one strain of mice could be transplanted to other individual mice within that strain, but not to mice of a different strain, who would reject them. Gorer, in 1937 (9), used similar experiments to demonstrate that the rejection of a tumour inherent to one mouse strain by mice of another strain was due to tissue antigen(s) (designated by Gorer as antigen II). This could raise tumour-destructive antibodies that would also agglutinate red cells of the tumour strain of mice.
The definitive analysis of the immunological nature of graft rejection was provided by Medawar (Fig. 9.3) and his co-workers. The need to treat severe burns incurred by both service personnel and civilians during World War II prompted the British government to sponsor a study of skin grafting. Medawar, in conjunction with a plastic surgeon, studied the use of skin autografts and homografts for burns. Initially both grafts seemed to take, but after a few days the allograft began to darken and die. Further allografts from the same donor were rejected even more rapidly. This became known as the “second set phenomenon” and was the observation that caused Medawar to conclude that the rejection of allografts was brought about by active immunisation (10).

Fig. 9.3 Sir Peter Medawar, painting by Sir Roy Calne. All rights reserved.
On his return to Oxford University after the war Medawar continued with his classic experiments which ultimately resulted in his Nobel Prize of 1960. Medawar and his colleagues set up an animal model to examine the mechanism of rejection of skin grafts under tightly controlled conditions. He grafted skin between rabbits and established the control data of average times to rejection. Repeated grafts from the same donors were rejected much more quickly, whereas grafts from a second donor lasted as long as the initial grafts. Medawar thus had illustrated clearly the specificity of graft rejection. He also made careful histological examinations of the grafts and described their invasion by the white blood cells. This established beyond doubt the immunological nature of the rejection reaction (7).
Prevention of Rejection: An Impossible Goal?
During the 1930s the concept of genetic individuality had emerged. Loeb, in his book The Biological Basis of Individuality, stated that any transplant between individuals would inevitably fail, since there were differences in the genetic make-up of everyone. Twenty years later, Loeb was invited to an international conference on transplantation. He declined, since he considered that “the subject matter of the conference was useless to pursue and a waste of time because the goal was impossible”(11). Unfortunately such emphatic assertions tend to be recorded in perpetuity. Since today there are patients with kidney transplants of more than 20 years standing, this provides a lesson for those tempted to make definitive statements based on intuition rather than evidence.
Experimental work during the 1950s increased our knowledge of immune mechanisms and strengthened the view that the rejection reaction might be capable of modification, so that transplants could succeed.
The fact that two genetic types could coexist in the same organism, without rejection of either, emerged from studies in cattle twins. Twinning is rare in cattle, and identical twinning is virtually unknown. In a study of cattle twins, Owen (12) noted that each twin carried blood cells of two different types. The fact that each was tolerated was presumed to be the result of an admixture of bloods between the twins in utero (Lillie, in 1916, had shown that in bovine twins there was often a union of the circulatory systems between the two placentae) (13).
The inevitable conclusion of these observations was that exposure to genetically dissimilar cells during fetal life rendered cells of that type tolerable in later life.
Other animal experiments consolidated this conclusion. Cannon and Longmire (14) transplanted skin between newly hatched (one-day-old) chicks of different breeds, producing adult red hens with patches of skin growing white feathers (in later experiments they also reversed the position of the skin, so that the white feathers were projecting in a different direction to the rest of the plumage). Delaying the skin graft for three days reduced the success rate to 1%, at 14 days all grafts were rejected.
Medawar also experimented with younger and younger animals, and in 1953 published a significant paper describing what he termed as “actively acquired tolerance” (as opposed to actively acquired immunity) (15). Medawar and his co-workers anaesthetised a mouse (CBA strain), pregnant and near to term. By a careful incision they exposed the abdominal body wall. The fetuses were sequentially brought into view by gentle manipulation of the abdomen and injected with a suspension of cells from adult mice of a different strain (A-line). Eight weeks after birth, each member of the CBA litter received a skin graft from an A-line mouse. The grafts took satisfactorily and resulted in the much-photographed mice carrying patches of fur of an entirely different colour. Medawar had thus shown that exposure of mice in utero to tissue of a genetically different strain produced a tolerance to that tissue.
Although “actively acquired tolerance,” as experimentally accomplished by Medawar, had no immediate application to the problem of rejection of kidney grafts, this work demonstrated that the immunological barrier was not insuperable. This was immensely encouraging for the surgeons endeavouring to achieve long-term survival of transplanted kidneys. (One such surgeon recorded his debt to Medawar in his account of the history of organ transplantation (16)).
The Identical Twins: A Vindication of the Surgery
It was evident that rejection of grafts would be unlikely if the organ or tissue came from an identical twin, since such monozygotic twins were also genetically identical. As everyone possesses a pair of kidneys, the surgeons at the Brigham Hospital had long considered that the donation of one kidney from a healthy twin might well be life-saving for the other suffering from CRF. The opportunity to put this to the test arose in 1953, when “Mr R H” was admitted to the Boston Public Health Service Hospital with chronic nephritis. His illness was severe and the outlook poor. Upon learning that the patient had a twin brother, his physician contacted the Brigham Hospital to suggest that this might be a case for a successful transplantation. Having ascertained that the tissue of the twins was interchangeable (by cross grafting patches of skin), and after establishing that the potential donor had a healthy urinary system, the question as to whether the transplant should be attempted was seriously addressed by the scientific and surgical team at the Brigham.
At a meeting on 20 December 1954 it was decided to proceed with the operation and, in view of Doctor Murray’s experience with dogs, to place the donated kidney in the abdomen with the ureter anastomosed directly to the bladder (7).
The operation was performed on December 23, 1954. The transplanted kidney began to produce “crystal clear normal urine” before the surgeons even had time to sew the severed ureter to the bladder. The patient recovered rapidly after the operation. His heart, which had been enlarged, returned to normal size, fluid cleared from his lungs and he was returned to health. Mr RH married the nurse who had tended him in hospital and raised a family.
To attempt this first identical twin transplant was a brave action by the medical team at the Brigham. It showed unequivocally that in man, as in dog, this operation was feasible and that the transplanted organ performed normally. Attention then turned to the apparently insuperable problem of maintaining grafts from genetically different donors.
Intense radiation was known to be particularly toxic to the mediators of rejection, the white blood cells. In view of the implacability of the problem it was not surprising that even this drastic procedure was examined as a means of prolonging survival of grafted kidneys. High levels of x-radiation were shown to prolong the life of skin homografts in rabbits, but at the cost of shortening the life of the host, since damage to the bone marrow rendered the animals susceptible to infection (17). Researchers tried to combat this problem by subsequent administration of homologous and heterologous bone marrow cells. The survival time of mice exposed to lethal x-radiation was increased by such treatment. Subsequently, skin grafts between rabbits showed extended survival after x-radiation and injection of hybrid bone marrow prior to grafting (18).
A successful renal graft was achieved in an irradiated dog at the Mary Imogene Bassett Hospital in Cooperstown. The animal was infused with bone marrow from the donor animal. Unfortunately the lack of an effective immune system resulted in the death of the animal from pneumonia after 49 days. However the grafted kidney functioned excellently for this period, with no sign of rejection (19).
The results of these animal experiments did not really augur well for the value of x-radiation in patients in need of a kidney graft. However, dire situations produce drastic action. X-radiation followed by administration of bone marrow from numerous relatives was used to prevent rejection of a kidney graft in a woman who had her only kidney removed to stem a life-threatening haemorrhage. After some while her platelet levels dropped and, perhaps predictably, she died of haemorrhage 32 days after the irradiation. Of great significance however, at the post mortem the kidney showed no sign of rejection (7).
Despite one apparent success, where the kidney was donated from a fraternal twin, and hence only a low level of radiation was used, the toxic effects of the radiation in patients, as in the animals, rendered this technique too dangerous. Nevertheless, the continued functioning of the transplanted kidneys did encourage the surgeons to believe that prevention of rejection was achievable (7).
Success with the First Immunosuppressants
A paper of great significance in transplant research is that of Robert Schwartz and William Dameshek of Tufts University Medical School. These workers showed, in 1959, that the anticancer drug 6-mercaptopurine (6-MP) given to rabbits, prevented the formation of antibodies following the injection of human serum albumin (20). In other words, 6-MP was capable of suppressing an immune response. Within one year Schwartz and Dameshek had extended this work to show that 6-MP could triple the survival time of skin homografts in rabbits (21).
The British surgeon, Roy Calne demonstrated the value of 6-MP in delaying the rejection of kidney allografts in dogs. In his first series of experiments, although 6 mg of 6-MP per day prevented rejection, the animals died within 10-14 days. In a second series, the animals were bilaterally nephrectomised (and thus had to depend solely on the graft) and were given 5 mg of 6-MP initially. This was reduced to 2.5 mg after 2 days. Incredibly, two dogs survived for 21 and 47 days respectively. Without 6-MP dogs so treated would survive only 4 to 8 days, and ultimately die of kidney failure. Both of the test dogs died of pneumonia. The kidneys, however, showed no sign of rejection and were secreting concentrated urine to the end (22).
Calne at this time joined the active team at the Brigham Hospital for a sabbatical, where a collaboration was formed with the synthetic chemists at the Burroughs Wellcome Research laboratories in Tuckahoe, New York. These researchers, under the leadership of Dr George Hitchings, synthesised new analogues of 6-MP and produced azathioprine, orally active and less toxic than 6-MP. With azathioprine it was possible to keep bilaterally nephrectomised dogs alive for up to 12 months (Fig. 9.4), dependent only on their grafted kidney (7). This experimental work led to the use of immunosuppressant drugs in patients with kidney grafts. From 1963 onward one-year survival of related donor transplants reached 80%.

Fig. 9.4 The first long-surviving dog, Lollypop, treated with the immunosuppressant azathioprine following a kidney graft. All rights reserved.
Corticosteroids, such as cortisone and the newer synthetic analogues, soon began to be used in conjunction with azathioprine. Corticosteroids were known to inhibit antigen-antibody reactions and were subsequently shown to produce a threefold increase in the survival time of dog kidney homografts (23).
In the late 1970s cyclosporin was extracted from the fungus Tolypocladium inflatum, and purified as a potential antifungal agent. It was however, shown to be rather better as a potent immunosuppressant in animal tests. It suppressed the appearance of plaque-forming cells in the spleens of mice immunised with sheep red blood cells, and doubled the survival time of skin allografts in mice. Cyclosporin has become established as a front-line immunosuppressant drug.
Since the lymphocyte is the cell primarily responsible for rejection, it is not surprising that antilymphocyte serum (ALS) has been developed as a potential antirejection agent. Metchnikoff in 1899 was the first to prepare ALS by injecting guinea pigs with extracts of rat or rabbit lymph nodes (25). The sera produced caused agglutination of rat or rabbit white cells. Woodruff clearly demonstrated the benefit of ALS in transplantation by showing it produced a ten-fold increase in survival time of skin homografts between albino and hooded rats (26). Despite problems of standardisation, ALS raised in horses is used in acute rejection crises. Poor tolerance to horse proteins, manifested by some patients, necessitates the occasional use of ALS raised in rabbits.
Tissue Typing
The genetic basis of rejection had been exposed by the mouse tumour transplant experiments of Little and Tyzzer (vide supra). The use of inbred strains of mice differing at only a single genetic locus from the original strain (congenic strains) enabled Gorer and Snell to establish the genetic locus (the “H” locus) for transplantation antigens. This work on the murine counterpart of human leucocyte antigens (HLA) hastened the understanding of transplantation antigens and emphasised the need for adequate tissue typing to ensure the best chance of long-term survival of kidney grafts (27)
The Future
Despite the emphatic progress made in organ transplantation, there is still need for improvement in antirejection therapy. Tacrolimus (FK 506) was introduced in 1989 but seems to share the same toxic effects as cyclosporin. Using transplant models in rodents, dogs and monkeys various groups of researchers have produced many candidate antirejection drugs (28). It is estimated that at the moment there are 10 novel non-peptide drugs and 15 monoclonal antibodies in various stages of clinical and preclinical testing (28). From this research will no doubt come further, step-by-step improvements in immunosuppressive therapy.
The continual improvements in methods to prevent rejection have resulted in an increase in patients on the waiting list for a transplant. The shortage of donors has thus become the major problem, exacerbated by the fall in donors resulting from improved road safety measures.
This problem would be eased if kidneys from another species (xenotransplants) could be used. A colony of transgenic pigs has now been produced which possess human genes intended to protect the transplant against the acute, complement driven, rejection process that normally occurs with xenotransplants. The first transplant from a pig to a human will no doubt be attempted within the next decade.
About 2,000 patients per year receive a kidney transplant in the UK (Figs. 9.5 and 9.6). Over two-thirds of these can expect their grafts to survive for at least a decade (1). In his admirable history of tissue transplantation, Francis Moore (then Professor of Surgery at Harvard) stated that none of the advances in kidney transplantation could have occurred without the extensive research in animals (7). Any dispassionate historian would agree. Without the pioneering work on anastomoses by Carrel, the early experiments on graft placement by Murray, together with the experimental trials with antirejection drugs, we would still today regard the diagnosis of CRF as a death sentence.
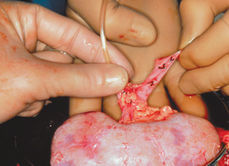
Fig. 9.5 A donor human kidney is perfused with saline prior to transplantation.
© Science Photo Library, all rights reserved.
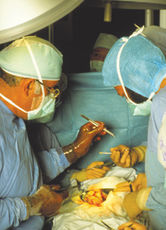
Fig. 9.6 Surgeons performing a kidney transplant operation. Of the 2,000 patients a year in the UK who receive a transplant, two thirds can expect their graft to survive for at least 10 years (Ref. 1). © Science Photo Library, all rights reserved.
Three Nobel Prizes have been awarded (Carrel in 1912, Medawar in 1960 and Murray in 1990) for work that has assisted in the success of transplantation. Many might argue that even more scientists in this field merit such recognition.
ANIMAL EXPERIMENTS AND KIDNEY TRANSPLANTATION
|
1902 |
||
|
1950 |
Autotransplantation of kidney Perfection of placement surgery |
|
|
1953 |
“Actively acquired tolerance” |
|
|
1955 |
Immunosuppressant effect of high dose radiation |
|
|
1959 |
Immunosuppressant drugs |
Rabbit, dog |
|
1963 |
Antilymphocyte serum |
|
|
1965 |
Tissue typing |
Mouse |
|
1989 |
An earlier version of this chapter was published as: The contribution of animal experiments to kidney transplantation. RDS News April 1995 8-14.
References
- West R (1991) Organ Transplantation. London: Office of Health Economics.
- Calne R (1970) A Gift of Life: Observations on Organ Transplantation. New York: Basic Books.
- Carrel A (1902) La technique operatoire des anastomoses vasculaire et la transplantation des visceres. Lyon Med 98 859.
- Carrel A (1912) Technique and results of vascular anastomoses. Surg Gynec Obst 14 246.
- Carrel A (1910) Remote results of transplantation of the kidney and spleen. J Exper Med 12 146.
- Williamson C (1926) Further studies on the transplantation of the kidney. J Urol 16 231.
- Moore F (1964) Give and Take: The Development of Tissue Transplantation. New York: Saunders.
- Little C & Tyzzer E (1916) Further experimental evidence on the inheritance of susceptibility to a transplantable tumour, carcinoma (JWA) of a Japanese Waltzing mouse. J Exp Res 33 393.
- Gorer P (1937) The genetic and antigenic basis of tumour transplantation. J Path Bact 44 691.
- Gibson T & Medawar P (1942) The fate of skin homografts in man. J Anat 77 299
- Murray J (1982) Reminiscences on renal transplantation, in Chatterjee S N (ed.)Organ Transplantation. Boston: John Wright.
- Owen R (1945) Immunogenetic consequences of vascular anastomoses between bovine twins. Science 102 400.
- Lillie F (1916) The theory of the free martin. Science 43 611.
- Cannon J & Longmire W (1952) Studies of successful skin homographs in the chicken. Ann surg 135 60.
- Billingham R, Brent L & Medawar P (1953) “Actively acquired tolerance” of foreign cells. Nature 172 603.
- Calne R (1970) Dedication in A Gift of Life. Observations on Organ Transplantation. Aylesbury: MTP.
- Dempster W, Lennox B & Boag J (1950) Prolongation of survival of skin homografts in the rabbit by irradiation of the host. Brit J Exp Path 31 670.
- Main J & Prehn R (1955) Successful skin homografts after administration of high dosage x-radiation and homologous bone marrow. J Nat Cancer Inst 15 1023.
- Mannick J, Lochte H, Ashley T & Ferrebee W (1959) A functioning kidney homotransplanted in the dog. Surgery 46 821.
- Schwartz R & Dameshek W (1959) Drug-induced immunological tolerance. Nature 183 1682.
- Schwartz R, Dameshek W & Donovan J (1960) The effects of 6-mercaptopurine on homograft reactions. J Clin Invest 39 952.
- Calne R (1960) The rejection of renal homografts inhibition in dogs by 6-mercaptopurine. The Lancet 1 417.
- Baker R, Gordon R, Huffer J & Miller G (1952) Experimental renal transplantation: 1. effect of nitrogen mustard, cortisone and splenectomy. Arch Surg 65 702.
- Woodruff H & Burg R (1986) The antibiotic explosion, in Discoveries in Pharmacology vol. 3 ed Parnham M & Bruinvels Amsterdam: J. Elsevier.
- Metchnikoff E (1899) Etude sur la resorption des cellules. Ann Inst Pasteur 13 737.
- Woodruff M & Anderson (1963) Effect of lymphocyte depletion by thoracic duct fistula and administration of antilymphocyte serum on survival of skin homografts in rats. Nature 200 702.
- Brent L & Sells R (1989) Notes on the history of tissue and organ transplantation, in Chatterjee SN (ed.), Organ Transplantation. Current Clinical and Immunological Concepts. Boston: John Wright.
- Maggon K (1994) Immunosuppressive gold rush and drug development. Drug News and Perspectives 7 389.
1 One surprising example of a homotransplanted kidney surviving for 175 days without any immunosuppressive therapy is described by Moore (The case of “Dr. W” in Ref. 7). The patient ultimately died as a result of severe hypertension, the kidney showing no classical sign of rejection. There is no explanation for this random success. It was an early example of the occasional apparent acceptance of a graft that occurs in both animal and human experiments, perhaps due to a chance close tissue matching.







