19. The Ethics of Maggot Therapy
© 2022 Frank Stadler, CC BY-NC 4.0 https://doi.org/10.11647/OBP.0300.19
Maggot therapy needs a social licence, which means that regulators, healthcare administrators, doctors, nurses, allied health providers, and patients must accept and support the treatment. Therefore, medicinal maggot production and maggot therapy must be informed and guided by strong animal and healthcare ethics. The first part of this chapter explores the animal ethics of rearing flies in laboratories and using medicinal maggots for wound care. The second part is dedicated to the biomedical and healthcare ethics of maggot therapy. Rather than a definitive treatise, this chapter should be understood as a first-pass examination of ethical issues related to maggot therapy.
Introduction
Maggot therapy is the treatment of wounds that require debridement and infection control with living fly larvae (maggots). Medicinal maggots are generally produced under strict quality control in medical laboratories and insectaries. Maggot therapy has great potential to be simple, affordable and highly efficacious in both high- and low-resource healthcare settings alike. Apart from supply-chain barriers hindering global supply, there is also the need for a social licence, which means that regulators, healthcare administrators, doctors, nurses, allied health providers, and patients must accept and support the treatment. Therefore, medicinal maggot production and maggot therapy must be informed and guided by strong animal and healthcare ethics, but any meaningful discussion of the ethical questions emerging from the use of medicinal maggots for healthcare purposes has not taken place, yet.
This paucity of work may be seen as a reflection of the status of invertebrates before the law as well as in public perception, and the fact that maggot therapy is just one of many healthcare interventions guided by ethical norms and local rules and regulations. In other words, biomedical ethicists and maggot therapy researchers may consider it already covered by discussions in general medical ethics. In addition, workers in the field of maggot-assisted wound care have been preoccupied with the biological-, biomedical-, and clinical issues that maggot therapy presents and may not have felt the need to engage on a scholarly basis with relevant ethical considerations. Moreover, foundational ethical principles in healthcare such as ‘do no harm’ are implicitly adopted in maggot therapy treatment guidelines that, for example, explain indications, contra-indications, side-effects, and risks [1]. Similarly, a discussion of maggot therapy in the context of humanitarian ethics has not been necessary because maggot therapy has not been part of any humanitarian response to disaster or war, yet.
This chapter is divided into two parts. The first explores the animal ethics of rearing flies in laboratories and using medicinal maggots for wound care. The second part is dedicated to the human ethics of maggot therapy within the context of biomedical and healthcare ethics. In most jurisdictions, flies and other invertebrates are not considered animals capable of suffering to the same extent as vertebrate animals. This has implications for the care and treatment of flies during mass rearing and maggot therapy, and particularly the disposal of maggots after therapy. From a healthcare ethics standpoint, the treatment of patients with maggot therapy should always be in line with international medical codes and declarations [2] and the four principles of biomedical ethics: autonomy, beneficence, nonmaleficence, and justice [3].
Rather than a definitive treatise on the ethics of maggot therapy, this chapter should be understood as a first-pass examination of ethical considerations as they apply to maggot therapy.
Animal Ethics and the Humane Treatment of Medicinal Maggots
Very little is known about an insect’s inner life, its ability to feel pain and distress, and whether this can be compared with our own human experiences of pain and distress. There is also very little information on methods for euthanasia of invertebrate animals and what method is the most humane for insects. There is, however, no doubt that medicinal maggot producers and maggot therapists have a moral obligation to minimise the potential suffering of these animals during production, distribution, and therapy.
Animal Ethics and Invertebrates: Are Flies Animals before the Law?
It is incumbent on anyone who utilises animals, for example for food, fibre, research, or companionship, to ensure the wellbeing of these animals. Medicinal flies are vulnerable to ill treatment from the time they are collected in the field to the time they are disposed of in the insectary, laboratory, or the clinical setting. It is therefore regrettable that insects are not considered animals before the law in Australia, the United States [4, 5], and other jurisdictions. The 8th edition of the Australian code for the care and use of animals for scientific purposes [5] defines animals as “any live non-human vertebrate (that is, fish, amphibians, reptiles, birds and mammals encompassing domestic animals, purpose-bred animals, livestock, wildlife) and cephalopods” (p. 3). The U.S. Animal Welfare Act, 1966, is even more restrictive regarding the animals falling under its purview. The ARRIVE guidelines [6], however, consider laboratory animals as “any species of animal undergoing an experimental procedure in a research laboratory or formal test setting.” (p. 258). An excellent and comprehensive review of ethical considerations regarding the use of insects in research has been prepared by Freelance [7]. It has been used to inform and structure the animal ethics discussion in this chapter.
Considering the healing benefits derived from medicinal maggots, it is fair to demand that their production, use, and eventual disposal should be guided by ethical principles that align with those applied to the treatment of vertebrates. Given the special ecological, anatomical, and physiological differences between vertebrates and calliphorid flies, some concessions will be reasonable, but there is very little literature on appropriate and humane treatment of invertebrate animals [8].
What Does the Ethical Utilisation of Medicinal Flies Look Like?
Freelance [7] made recommendations for ethical research with insects which also apply to medicinal maggot production, maggot therapy research, and maggot therapy itself (Table 19.1). It appears prudent to use these recommendations as the scaffold for the discussion of what humane and ethical utilisation of medicinal flies may look like.
Table 19.1 Recommendations by Freelance [7] for the ethical treatment of insects in research, applied to medicinal maggot production and maggot therapy.
|
Recommendation (abbreviated) |
Recommendation applied to the treatment of medicinal flies |
|
Responsible research design |
Experimentation with live medicinal flies of all life stages should be designed and conducted to optimise data quality and minimise the impact on the experimental animals. This intersects with the reduction and refinement recommendations below. |
|
Beneficence |
The benefit of treating wounds with medicinal maggots should outweigh the harm done to flies during production and to medicinal maggots in maggot therapy. |
|
3Rs — Replacement |
Medicinal maggots should be replaced, where possible, with other wound care therapies that do not use living animals. |
|
3Rs — Reduction |
The number of flies kept to maintain colonies and to produce medicinal maggots should be minimised to the amount that is necessary and overproduction should be avoided. This also applies to the production of medicinal maggots for therapy. |
|
3Rs — Refinement |
Husbandry practices, medicinal maggot production methods, and therapy procedures should be developed and refined so that they minimise the potential suffering of flies and maggots. |
|
Support for natural behaviours in captive specimens |
Conditions for natural behaviour of flies and maggots should be provided in the insectary, e.g. sufficiently large cages for flight. |
|
Responsible and sustainable live animal capture |
Medicinal flies for the establishment or genetic replenishment of fly colonies in the laboratory should be collected in a way that minimises harm to the collected flies, non-target species, or the ecosystem. Moreover, meat bait for attracting flies should be sourced from already butchered animals or road kill. |
Responsible research design. Much of what is known of medicinal maggots regarding their ecological, life-history, and physiological characteristics, and clinical performance has been learned from experiments and trials. Such research should be designed and executed so it produces high-quality data, minimises the number of flies used (all life stages), and minimises the negative impact of experimental treatments on the animals.
The idea of responsible design can also be extended to the design of the maggot-therapy supply chain. For most aspects of fly colony maintenance, medicinal maggot production, distribution, and therapy, the objectives of animal welfare align with the objective to produce high-quality, healthy medicinal maggots. For flies to produce large numbers of eggs for the production of medicinal maggots, they need to be in good health and receive optimal nutrition. At the distribution stage of the maggot therapy supply chain the producer is obliged to package medicinal maggots in protective primary and secondary packaging and then to transport the consignment in insulated transport shippers, which minimises mortality and ensures that healthy, viable maggots reach the point of care [9, 10]. The dressing technology for the application and retention of maggots to the wound, as well as patient instructions and care directives during treatment, all ensure that maggots find conditions for rapid and optimal debridement and thus growth [11]. It is only at the end of treatment that the maggots will need to be disposed of in the clinical waste stream, as will be discussed in more detail later.
Beneficence. The ethical principle of beneficence says that on balance any intervention should be beneficial by maximising the benefit and minimising harm as much as is possible [3]. This principle can be applied to both animal ethics and human ethics, as will be discussed later. Here, the focus is on the benefit of maggot therapy to the patient versus the harm it causes to medicinal flies. Maggot therapy is a highly efficacious wound care therapy and its clinical benefits [12, 13] as well as its therapeutic principles [14–16], have been presented in this book. With this in mind, it can be argued that the sacrifice of flies that have been sustainably reared in the insectary rather than collected from the wild is justified.
It also helps to consider the ecological and evolutionary strategy calliphorid flies pursue. They are an r-selected species and their reproductive strategy is such that they produce large numbers of eggs in order to capitalise on ephemeral resources and high mortality of offspring [17]. Each female may lay 1000 to 2000 eggs in her lifetime [18, 19]. If all offspring survive, they would exponentially multiply. However, the world is not drowning in flies, owing to high mortality of flies during all life stages. Indeed, the vast majority of offspring succumb to unfavourable climatic conditions, disease, or become prey before they can reproduce. This ecological strategy provides a strong ethical rationale for the sustainable utilisation of a species such as the greenbottle blowfly, Lucilia sericata, whose evolutionary destination is to naturally sacrifice most of its offspring to predation and other misfortune. Some may suggest that the ecologically-determined and habitually-experienced misfortune of flies in the wild should not be justification for equal treatment in captivity. The difference here is that the domestication and exploitation of flies may well be justified on ecological grounds, but not their ill treatment. As will be discussed later under ‘Refinement’, it is imperative that any potential suffering of medicinal flies is minimised or avoided altogether.
3Rs — Replacement. Over the past decade, there has been considerable interest in the excretions and secretions of medicinal maggots. This research has revealed that the therapeutic benefits of debridement, infection control, and wound healing are brought about by enzymes and other biochemical factors secreted or excreted by the maggots [14–16]. For example, in 2017 the company SolasCure was founded to develop and commercialise a hydrogel dressing containing an enzyme found in medicinal maggot excretions and secretions [20]. While this is in principle a welcome step from an animal ethics standpoint, it is not certain whether a drug based on one enzyme only has the same therapeutic effect as is provided by the whole organism. This means the benefit derived from maggot therapy may continue to outweigh the harm caused to the few medicinal maggots used for treatment. Secondly, drug development is expensive and developers want to recoup these costs plus profit. Therefore, it is unlikely that disadvantaged and impoverished populations around the world will be able to afford such innovative cures until generic versions become available after the patent has expired. In the meantime, there is a strong argument for the use of affordable maggot therapy to treat wounds, particularly in low- and middle-income countries.
3Rs — Reduction. The best way to minimise potential suffering is to avoid it altogether. Flies are prolific breeders with each female laying up to 2000 eggs over her lifetime. This means that there is the potential to breed enormous numbers of flies. However, producers should only maintain enough flies to be able to meet unforeseen demand for medicinal maggots, to maintain genetically diverse colonies, and perhaps for research purposes. Producers will also be inclined to maintain only necessary fly stock for financial reasons, i.e. to reduce variable costs such as labour and consumables. To conclude, by maintaining a stable population of flies with excess individuals produced being diverted to medicinal maggot production, the producer mimics natural population-limiting ecological dynamics.
3Rs — Refinement. Pain, distress, and general suffering should be minimised when collecting, transporting, captive breeding, and euthanising animals. The objectives for each are very similar but it is instructive to review those for euthanasia in more detail. According to Close and colleagues [21] the method for killing an animal should be
- painless
- quick
- minimally distressing to the animal
- reliable
- reproducible
- irreversible
- simple to administer
- safe for the operator
- aesthetically acceptable.
The knowledge base for humane treatment of invertebrates in zoos, research, and elsewhere is still poor and there is, as yet, an absence of reliable knowledge as to whether invertebrate animals perceive pain the same way vertebrate animals and humans do [22], let alone what their psychological states might be under certain circumstances. Nonetheless, mounting evidence reviewed by Freelance [7] suggests that some insects, for example, may experience emotive states similar to vertebrate animals. As for euthanasia and analgesia of invertebrate animals, the Institute for Laboratory Animal Research has published a special edition dedicated to invertebrate animals. It recommends anaesthesia for terrestrial invertebrates using isoflurane (5–10%), sevoflurane, halothane (5–10%), or carbon dioxide (CO2; 10–20%), and for aquatic invertebrates tricaine (Sigma) and benzocaine [22, 23].
- Euthanasia in the fly insectary. The health of flies that reach the end of their productive lifetime may slowly decline in the absence of predators who would ordinarily put a swift end to any unfit prey. When productivity declines and age-related mortality increases, it is time to terminate the colony. Current practice (author’s) is to euthanise adult flies still in their cage (assembled or collapsed) at -20℃. When harvesting eggs, any surplus not used for medicinal maggot production and colony propagation should also be placed in the freezer while still at the egg stage.
- Euthanasia in the laboratory. Surplus disinfected maggots and pre-packaged maggots for shipment that are not sent out to clinicians should also be euthanised as soon as feasible by freezing.
- Disposal after therapy. It is at the point of care where animal welfare considerations are hardest to observe because healthcare providers are time-poor, must prioritise the welfare of patients, and observe infection control practices. The Joanna Briggs Institute [24] recognised in their report, Recommended Practice: Wound Debridement–Larval Therapy, the need for humane treatment of medicinal maggots and suggested that practitioners should place used medicinal maggots in the freezer to kill them prior to disposal. Whether healthcare providers are willing to put soiled dressings and maggots into shared clinic freezers, double-bagged or not, is another matter.
When studying the development times of forensically important blowflies, some researchers use alcohol to kill the maggots ahead of measurement [25] and others kill maggots swiftly by immersion in hot water [26]. When considering the speed with which each of the currently practiced euthanasia methods work, immersion in hot water is by far the fastest, resulting in instant death, followed by freezing, which may take a few minutes depending on how exposed the eggs, maggots, and adult flies are. Cooling slows down cold-blooded animals’ metabolism until sub-zero temperatures cause tissue freezing and death. This is a fairly rapid process in small animals with a large surface area such as maggots. Immersion in 70% ethanol is by far the slowest method and maggots may still be alive after an hour of immersion (author’s observation), which suggests that it is the lack of oxygen that kills the maggots eventually rather than the alcohol. It must be stressed that immersion in hot water or pouring boiling water over soiled dressings and maggots after treatment is impractical and can be quite dangerous. Handling boiling water is dangerous in and of itself but in addition, germs and toxins may become airborne with the steam that is generated and may lead to serious health problems when breathed in—a phenomenon that has been documented for aquarists who used boiling water to sterilise marine rocks [27].
For now, given insufficient research and practical solutions for euthanasia of maggots, the safest bet is to freeze medicinal maggots prior to disposal. If maggots are disposed of in the clinical waste stream, they will end up being incinerated or otherwise treated with heat, chemicals, or irradiation [28]. Industrial clinical-waste incineration facilities operate at very high temperatures and death should come almost instantaneously to disposed maggots. Incinerators used in low- and middle-income country healthcare settings are often less efficient and operate at lower temperatures. In such cases, it can be assumed that the burning of waste and therefore the euthanasia of maggots is a slower process leading to a more gradual heating of maggots among other clinical waste. Of course, maggots may also succumb long before incineration due to crushing during handling, a build-up of CO2 in the waste bag, and contact with other healthcare chemicals in the same waste bag.
Support for natural behaviours in captive specimens. There is a trade-off between production efficiency and practicality on one hand, and environmental enrichment for the benefit of flies on the other. It is unrealistic to provide life-like vegetated aviary enclosures for flies that would simulate the natural environment. A suitable compromise is to provide roomy insect cages. In the author’s experience, cages of 450 x 450 x 450 mm provide ample room for flight. It is also advisable to illuminate the insectary with daylight-spectrum lighting for optimal fly health. Adult flies are usually dispersed in the ecosystem and congregate in large numbers only when feeding and laying eggs on cadavers or as larvae feeding on such cadavers. Although high density of flies can lead to aggression, particularly by males toward females, it is in the interest of the producer to maximise efficiency and therefore the stocking rate of flies in insectary cages. Producers are encouraged to monitor colony performance and health regularly and adjust stocking rates as necessary. In the author’s experience, stocking rates of up to 1 fly per 110 cm3 are sustainable and do not lead to high or premature mortality, provided all other environmental requirements are met. Please refer to Chapter 14 for a comprehensive discussion of medicinal fly husbandry [29].
Optimising fly wellbeing through the provision of optimal environmental conditions that stimulate natural behaviour is not only an ethical imperative but it also makes good business sense. Any practice that improves the wellbeing of flies in the production system will translate into a better product, i.e. medicinal maggots for clinical treatment. In addition, if the humane treatment of flies can be demonstrated to clients it may also increase consumer acceptance, as is the case in the poultry industry, where free-range and barn-laid eggs are preferred by ethically-minded consumers.
Responsible and sustainable live animal capture. Producers and researchers may collect flies to establish colonies or to replenish genetic material after longer periods of captive breeding. All life stages may be collected. The recommendation for responsible and sustainable live animal capture (Table 19.1) requires collectors to reduce bycatch when traps are used. This may be difficult as a range of fly species in addition to the target species may be trapped. Bycatch might be reduced by monitoring catch rate and not leaving the trap set up for longer than necessary. Fortunately, the aim is to capture live flies so when sorting takes place, non-target species can be released if practical. Bycatch of adult flies can be altogether avoided if immature life stages (eggs and maggots) are collected from cadavers or putrid meat bait is offered to attract flies for oviposition. When transporting captured specimens to the insectary, it is in the interest of the collector to store them in an insulated transport container to avoid overheating. Finally, there is generally no need to purposely kill a domestic or other livestock to attract flies. Road kill is usually not hard to find and inexpensive meat waste from butchers is also suitable for attracting flies. Chapter 13 provides guidance on the establishment of laboratory colonies including the collection of fly breeding stock.
The Healthcare Ethics of Maggot Therapy
Having reviewed the ethics of medicinal maggot production and maggot therapy from an animal ethics standpoint, we now shift to investigate the human ethics of maggot therapy, particularly in compromised healthcare settings such as in war, disaster, poverty, or extreme geographic isolation. Here, the focus shifts from medicinal flies to the relationship between care provider and patient—before, during, and after maggot therapy. The ethical foundations for this relationship have already been laid. There are a plethora of ethical codes and declarations relevant to healthcare delivery. Amnesty International has compiled most of them in the 5th edition of Codes of Ethics and Declarations Relevant to the Health Professions [2]. Some of these are of particular interest to medical practitioners who provide wound care in ordinary times and during war, disaster, and development:
- The Hippocratic Oath
- WMA Declaration of Geneva
- International Code of Medical Ethics
- Declaration of Helsinki: Ethical Principles for Research Involving Human Subjects
- Regulations in Time of Armed Conflict
- Geneva Conventions
- Common Article 3 of the Geneva Conventions
- Convention I For the Amelioration of the Condition of the Wounded and Sick in Armed Forces in the Field. Geneva, 12 August 1949.
- Convention II For the Amelioration of the Condition of the Wounded and Sick and Shipwrecked Members of Armed Forces at Sea. Geneva, 12 August 1949.
The field of medical and healthcare ethics has been greatly influenced by Beauchamp and Childress’ seminal work on the principles of biomedical ethics, now in its 5th edition [3]. The four principles are autonomy, beneficence, nonmaleficence, and justice. Autonomy refers to the patients’ ability to make their own decisions in healthcare which includes the ability to give informed consent or refuse a particular intervention. Beneficence requires healthcare to be of benefit to the patient. In this respect there can be conflict between the interests of the individual patient and the larger patient cohort, especially when healthcare resources are limited and utilitarian decisions have to be made to maximise the benefit to all patients. The principle of nonmaleficence asks the care provider to do no harm, while justice calls for the fair, appropriate, and equitable treatment of patients. Much of the discussion in this chapter is informed by Beauchamp and Childress [3] but this does not mean that the other codes of ethics listed in the Amnesty International compendium [2] can be ignored. Fortunately, they greatly overlap and align with the ethical notions encapsulated in the four principles while tailored to a particular healthcare setting, as the Geneva Conventions are tailored to armed conflict. A detailed discussion of the ethics of maggot therapy in all these contexts is beyond the scope of this chapter.
Autonomy
For clinicians who understand the therapeutic benefits of medicinal maggots and have successfully treated many wounds with maggot therapy, it may become somewhat tedious to sensitise and educate each new patient whose wounds are amenable to maggot therapy. However, there is no escaping this obligation. The principle of autonomy demands that patients have control over their bodies and are in the position to make treatment decisions.
Beauchamp and Childress propose a stepwise process by which the practitioner ensures that the patient understands what the treatment entails, consents to a treatment plan, and authorises this treatment plan [3, p. 80]:
I. Threshold Elements (Preconditions)
1. Competence (to understand and decide)
2. Voluntariness (in deciding)
II. Information Elements
3. Disclosure (of material information)
4. Recommendation (of a plan)
5. Understanding (of 3. and 4.)
III. Consent Elements
6. Decision (in favor of a plan)
7. Authorization (of the chosen plan)
Competence and voluntariness. In the first instance, the patient must be able to understand care information given by the practitioner and then able to make care decisions. If patients can’t make their own decisions (i.e. they are not autonomous), their interests need to be faithfully represented. Consider the following scenarios. A patient with a pressure ulcer might also have advanced dementia, as was the case with the author’s own grandfather, which means he is no longer able to make his own decisions. Another example might be found in a conflict or disaster setting, where the blast wound of a comatose civilian casualty has become severely infected with methicillin-resistant Staphylococcus aureus. In such cases when patients are unable to make their own decisions, Beauchamp and Childress [3] suggest that an advanced care directive by the patient is preferable. Advanced care planning seeks to clarify and capture how the patient would want to be treated, what treatments the patient would refuse, and who might be vested with the power of attorney to represent the patient’s wishes [30]. Such care directives may be of a general nature or more specific but it seems unlikely that patients would give directives explicitly on maggot therapy, perhaps unless they already face chronic wound care challenges. In the absence of advanced care plans, the decision should be made by a person who knows the patient well. This person should be able to make a decision that reflects the patient’s opinions and attitudes, in other words, a decision the patient would most likely have made. In cases where the patient was never autonomous or there is no representative such as a next of kin, and it is therefore impossible to get a sense of the patient’s wishes, a surrogate must decide in the patient’s best interest:
a surrogate decision maker must determine the highest net benefit among the available options, assigning different weights to interests the patient has in each option and discounting or subtracting inherent risks or costs. The term best is used because the surrogate’s obligation is to maximize benefit through a comparative assessment that locates the highest net benefit. The best-interests standard protects another’s well-being by assessing risks and benefits of various treatments and alternatives to treatment, by considering pain and suffering, and by evaluating restoration or loss of functioning. [3, p. 102]
Of course, the decision to accept or refuse maggot therapy must be voluntary and neither the practitioner nor other third parties must exert undue influence over the patient’s decision [31]. Given the unusual and for some people disconcerting nature of maggot therapy, there is always the danger that patients are unduly encouraged or dissuaded by their loved ones. Likewise, a practitioner who is passionate about maggot therapy may be tempted to pressure the patient to consent. Therefore, the role of objective sensitisation and education in maggot therapy cannot be overemphasised.
Disclosure, recommendation, and understanding. Practitioners need to consider what a reasonable person would want and need to know about maggot therapy. This typically includes information about the wound and its prognosis, the benefits of maggot therapy, alternative treatments, side-effects, risks, and how maggot therapy may limit daily life (e.g. bathing and showering, off-loading, etc.), and a treatment plan [32]. Fortunately, the general concept of maggot therapy and its three main therapeutic principles is easily understood especially when explained in simple, non-technical terms, and the same is true for the dressings used to apply and keep maggots in place. However, clinicians will need to tailor the information to the individual needs of the patient [3], especially because maggot therapy is still considered an unusual treatment. It is important to also take the patient’s attitude toward flies, maggots, and other invertebrate animals (‘creepy crawlies’) into account. While mere dislike of maggots and repulsion are not reliable predictors of non-consent, Petherick and colleagues suggest the use of positive language and phrases to describe the maggots’ therapeutic benefits [33]. Depending on the patient, it may also be helpful to explain the biology of maggots and how their evolutionary history and adaptation to unhygienic environments has given rise to the multiple therapeutic properties of maggot therapy.
Decision and authorisation. As the patient receives appropriate and easily understood information about maggot therapy and the treatment plan, the clinician needs to encourage patient feedback through open dialogue, and deduce whether the patient has understood maggot therapy, and what the consequences of consenting or declining the treatment would be [3]. Clinicians generally seek only verbal consent for relatively minor non-surgical procedures, which would include maggot therapy. However, depending on local rules and regulations and the patient’s capacity to make a decision, clinicians may need to obtain written consent or refusal of consent from the patient or surrogate [34].
Beneficence and Nonmaleficence
Medical treatments, including maggot therapy, must be performed with the primary objective to benefit the patient. At the same time, the treatment should not harm the patient. In practice, there are many treatments with varying efficacy and varyingly harmful side-effects. Thus, in order to align with the principles of beneficence and nonmaleficence it is necessary to maximise the net-benefit of any treatment regimen [35].
Chronic and infected wounds have been recognised as a major healthcare challenge in both high-resource and low-resource care settings such as disasters, conflict, and poverty [36–39]. Patients or their insurers in high-resource healthcare systems can afford advanced therapies and wound dressings but these are out of reach for people with wounds in low- and middle-income countries where a chronic wound can incur catastrophic healthcare expenditure for patients and their families [40]. Moreover, when limbs need to be amputated due to life-threatening infection this results in prolonged incapacity or even disability which greatly diminishes the patient’s ability to care for their families and contribute to society.
Maggot therapy is widely applicable and generally indicated for non-healing wounds with necrotic tissue and slough including infected wounds [41]. Maggot therapy can improve wound care particularly in low-resource healthcare settings because it not only removes necrotic tissue but also controls infection and promotes wound healing. This means that wound care with maggot therapy is less reliant on surgical expertise, antibiotic use, and advanced wound dressings [42]. It follows that on the health system- and societal level, the use of maggot therapy aligns with the principle of beneficence. As far as treatment decision making for individual patients is concerned, practitioners have to consider a range of factors that determine whether maggot therapy will on balance benefit the patient: i) wound aetiology and condition, ii) general patient characteristics and comorbidities, iii) treatment alternatives, iv) contraindications and treatment risks, and v) cost of treatment and the patient’s capacity to pay. As with all therapies and medicines, there are contraindications that increase the risk of adverse outcomes for the patient, such as allergic reaction, pain, or bleeding [1]. However, most of these contraindications can be carefully monitored and managed or circumvented, especially if the benefits of maggot therapy outweigh the risk of adverse events.
While informed consent of the patient is always necessary, the ultimate responsibility regarding patient safety still rests with the practitioner. This is not to say, though, that the producers and suppliers of medicinal maggots are absolved of responsibility. Indeed, in well-regulated healthcare systems the manufacturers of medical goods are required to adhere to stringent quality control standards and Good Manufacturing Practices [43]. If medicinal maggots are produced in low- and middle-income countries the sophistication of producers may vary greatly, especially because medicinal flies are very easy to keep and breed. In such circumstances it may be difficult to hold producers accountable for poor-quality medicinal maggots and adverse outcomes. However, there have been efforts by the author and colleagues to provide lay producers of medicinal maggots and healthcare providers in compromised healthcare settings with easy-to-understand visual, multi-lingual, and multimedia instructions to ensure production of high-quality medicinal maggots and safe maggot therapy (http://www.medmaglabs.com/creating-hope-in-conflict). Fortunately, adverse outcomes due to poor production practice is probably a rather small risk because of the extraordinary capacity of medicinal maggots to control infection even in cases of myiasis, the unintended wild colonisation of wounds with fly maggots [44]. In other words, as long as appropriate fly species are used to produce medicinal maggots, the risk to patients is limited. Nevertheless, it is incumbent on the treating practitioners to ensure that they procure medicinal maggots from reputable and proficient producers irrespective of the level of regulatory oversight in that healthcare system.
Justice
Maggot therapy in a resource-constrained healthcare setting. The moral obligation on healthcare providers to provide optimal care to every patient is at odds with the utilitarian imperative as it applies to resource-constrained healthcare settings. Under such circumstances, the aim is to provide the best possible wound care to the greatest possible number of patients, rather than prioritising optimal care to only a few patients [3]. This means that compromises need to be made in the care that can be provided (Figure 19.1). However, in care settings where low-cost medicinal maggots are made available, maggot therapy can ensure highly efficacious wound care for a large number of patients even if other wound care modalities, antibiotics, and medical personnel are in short supply. This is because maggot therapy i) can be performed by nurses rather than physicians, ii) can replace costly conventional wound dressings, iii) can replace, or reduce dependence on, the use of antibiotics, iv) can replace surgical debridement, and v) can promote wound healing and thus prepare wounds for surgical closure while patients in immediate and urgent need of care are looked after [45].
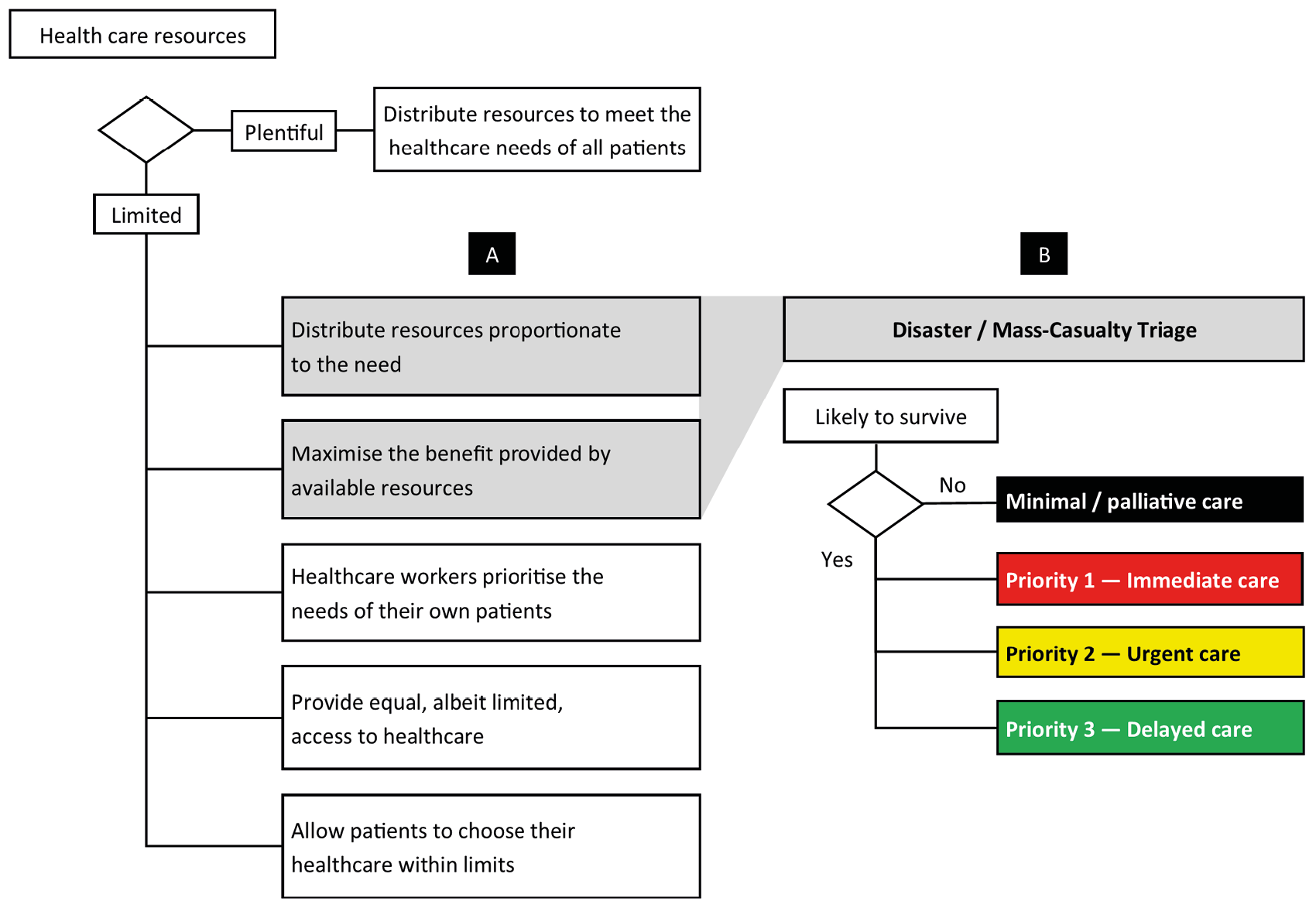
Figure 19.1 Abbreviated and simplified ethical framework for the provision of care in resource-constrained healthcare settings [35, 46]. Strategies for ethical healthcare provision in Column A may apply to any number of situations where resources are limited. However, the utilitarian approach of proportionate distribution that maximises benefit applies specifically to disaster triage (Column B). CC BY-NC.
Lack of access to limb- and life-saving treatment. Having pointed out that maggot therapy can make a meaningful contribution to fair healthcare provision in compromised healthcare settings, it is regrettable that maggot therapy has not yet been used in disaster settings and is unavailable in all but a few low- and middle-income countries. Despite the deep history of tribal and ancient use of maggot therapy in many regions of the world [47], maggot therapy is mainly offered to patients in North America, Europe and to a lesser extent elsewhere such as in Iran, Malaysia, Singapore, Japan, and Australia. With exceptions, these countries are generally high- or middle-income countries with excellent to reasonable access to healthcare and advanced wound care. In compromised healthcare settings, where maggot therapy could make a real difference, it is rarely used. There have been a number of attempts to introduce maggot therapy to Sub-Saharan Africa but such efforts have failed or are hampered by regulatory and bureaucratic barriers. For example, a recent programme supported by a grant from the Slovak government established a medicinal maggot production laboratory in Kenya and supported the training of the local workforce [48, 49]. Despite documented positive outcomes and improvements to the care of intractable wounds, delays in regulatory approval meant that the therapy could only be offered to very few patients in Nairobi and surrounding areas. A case report of the Kenyan maggot therapy programme is provided in Chapter 15 and with regard to distribution logistics in Chapter 17 of this book [10, 49].
As discussed under the principle of autonomy, patients are usually dependent on the expertise and advice given by doctors. In the case of maggot therapy this is no different. For the therapy to be widely adopted, it is necessary for doctors, nurses, and the greater healthcare system to endorse maggot therapy and to include it in their treatment ‘toolbox’. Prejudicial dislike or disregard for maggot therapy among healthcare professionals is still widespread, especially in jurisdictions where the therapy is new or has not been widely used in the past (personal observation). This, along with aggressive lobbying and marketing by manufacturers of competing wound care products, means that many patients miss out on affordable and highly efficacious maggot therapy. One might think that practitioners in low-resource healthcare settings would embrace maggot therapy but this is not necessarily the case. It is common in low- and middle-income countries that practitioners manipulate patients to make care decisions in which they have a financial interest [50]. In the case of chronic wound care, physicians may recommend surgical debridement instead of maggot therapy because the treatment incurs more surgeon time and therefore greater income [48]. Therefore, a highly affordable and efficacious therapy such as maggot therapy may struggle to gain wide-spread endorsement in favour of more lucrative treatment options. Key to overcoming these barriers are education campaigns for the public, patients, and healthcare professionals that translate and communicate the mounting clinical evidence in favour of maggot therapy. Winning the hearts and minds of future patients, doctors, and nurses may start as early as primary school when children have not yet formed strong negative opinions of maggots [51].
Supply-chain constraints are also responsible for lack of access to maggot therapy, especially in compromised healthcare settings. Medicinal maggots have a short shelf-life, requiring delivery at a temperature of 6–25OC and application to the wound within 24 to 48 hours of dispatch from the laboratory [52]. During times of disaster, in conflict, or in impoverished regions of the world such rapid and reliable distribution of goods is not possible because of broken and disrupted logistics infrastructures. However, these logistics barriers may be overcome with drone transport [53] and innovative supply-chain solutions that locate medicinal maggot production at the point of care. The latter may be achieved, for example, with mobile laboratories for medicinal maggot production, and through capacity building of isolated communities and medics working in austere care settings [54, 55]. This is because medicinal maggot production is a relatively simple process and does not necessarily depend on high-tech equipment or special expertise. Production and supply may be taken up by communities or entrepreneurs that seek an income from breeding and selling medicinal maggots. This economic incentive may further motivate service delivery even in austere settings. However, there is also a chance that treatment becomes unaffordable to the very poor, thus reinforcing healthcare injustices. Such entrepreneurs should be given advice on business models that allow producers and practitioners to cross-subsidise the treatment of the poor through charging wealthy patients higher fees.
Summary
This chapter concludes the present book on maggot therapy but it could have easily been the introductory chapter. Ethical considerations are central to all aspects of healthcare service delivery. Through this lens, maggot therapy can be seen as one of the countless medicines, devices, and therapies that are equally subject to ethical standards. However, the use of living organisms in place of inanimate drugs and devices sets maggot therapy apart from other wound care therapies. Maggots may conjure up feelings ranging from fascination to discomfort or even disgust. Therefore, to appropriately seek true, informed consent, providers of maggot therapy must pay closer attention to patient sensitisation and education. Moreover, the use of living organisms as a therapy adds another layer of complexity. In addition to regular biomedical ethics along the lines of the four principles discussed above, there are animal ethics issues that demand consideration. Unfortunately, little is known about the capacity of invertebrate animals such as flies and their larvae to experience distress and pain, but the question of suffering is central to animal ethics. Consequently, it is incumbent on maggot producers and therapists to be precautious and minimise any potential human and insect suffering.
Even with the best intentions, producers and clinicians may need to compromise on ethics because healthcare professionals are not always completely free to practice according to best-practice ethical standards and principles. Health and safety laws and regulations, and health service providers guided by religious faiths may take a different view on ethical conduct [56]. For example, a Jain patient may object on religious grounds to the killing of a living being such as a maggot [57]. The solution to this could be releasing used maggots after maggot therapy, rather than disposing of them along with the dressing waste. However, for public health and practicality reasons, this is clearly not possible. Wound care waste is considered infectious waste because soiled dressings, including medicinal maggots, are usually contaminated with pathogens. Therefore, this waste must be collected and disposed of safely, which invariably leads to maggots being killed in the process.
These limitations highlight the need for greater consideration of ethical issues in maggot therapy. It is my hope that both practitioners and scholars are encouraged to engage in this debate and eventually contribute to the development of ethical methods and practices in medicinal maggot production and maggot-assisted wound care.
References
1. Mexican Association for Wound Care and Healing. Clinical Practice Guidelines for the Treatment of Acute and Chronic Wounds with Maggot Debridement Therapy. 2010. https://s3.amazonaws.com/aawc-new/memberclicks/GPC_larvatherapy.pdf.
2. Amnesty International, Codes of Ethics and Declarations Relevant to the Health Professions: An Amnesty International Compilation of Selected Ethics and Human Rights Texts and Other Standards. 2009, London: Amnesty International.
3. Beauchamp, T.L. and J.F. Childress, Principles of Biomedical Ethics. 2001, New York: Oxford University Press.
4. Garber et al., Guide for the Care and Use of Laboratory Animals, Eighth Edition. 2010, Washington, D.C.: The National Academy Press.
5. National Health and Medical Research Council, Australian Code for the Care and Use of Animals for Scientific Purposes, 8th edition. 2013, National Health and Medical Research Council: Canberra.
6. Kilkenny, C., et al., Improving Bioscience Research Reporting: The ARRIVE Guidelines for Reporting Animal Research. Osteoarthritis and Cartilage, 2010. 20(4): pp. 256–260, https://doi.org/10.1016/j.joca.2012.02.010.
7. Freelance, C.B., To Regulate or Not to Regulate? The Future of Animal Ethics in Experimental Research with Insects. Science and Engineering Ethics, 2019. 25(5): pp. 1339–1355, https://doi.org/10.1007/s11948-018-0066-9.
8. Stadler, F., The Maggot Therapy Supply Chain: A Review of the Literature and Practice. Med Vet Entomol, 2020. 34(1): pp. 1–9, https://doi.org/10.1111/mve.12397.
9. Stadler, F., Packaging Technology, in A Complete Guide to Maggot Therapy: Clinical Practice, Therapeutic Principles, Production, Distribution, and Ethics, F. Stadler (ed.). 2022, Cambridge: Open Book Publishers, pp. 349–362, https://doi.org/10.11647/OBP.0300.16.
10. Stadler, F., Distribution Logistics, in A Complete Guide to Maggot Therapy: Clinical Practice, Therapeutic Principles, Production, Distribution, and Ethics, F. Stadler (ed.). 2022, Cambridge: Open Book Publishers, pp. 363–382, https://doi.org/10.11647/OBP.0300.17.
11. Sherman, R., Medicinal Maggot Application and Maggot Therapy Dressing Technology, in A Complete Guide to Maggot Therapy: Clinical Practice, Therapeutic Principles, Production, Distribution, and Ethics, F. Stadler (ed.). 2022, Cambridge: Open Book Publishers, pp. 79–96, https://doi.org/10.11647/OBP.0300.05.
12. Sherman, R., Indications, Contraindications, Interactions, and Side-effects of Maggot Therapy, in A Complete Guide to Maggot Therapy: Clinical Practice, Therapeutic Principles, Production, Distribution, and Ethics, F. Stadler (ed.). 2022, Cambridge: Open Book Publishers, pp. 63–78, https://doi.org/10.11647/OBP.0300.04.
13. Sherman, R. and F. Stadler, Wound Aetiologies, Patient Characteristics, and Healthcare Settings Amenable to Maggot Therapy, in A Complete Guide to Maggot Therapy: Clinical Practice, Therapeutic Principles, Production, Distribution, and Ethics, F. Stadler (ed.). 2022, Cambridge: Open Book Publishers, pp. 39–62, https://doi.org/10.11647/OBP.0300.03.
14. Nigam, Y. and M.R. Wilson, Maggot Debridement, in A Complete Guide to Maggot Therapy: Clinical Practice, Therapeutic Principles, Production, Distribution, and Ethics, F. Stadler (ed.). 2022, Cambridge: Open Book Publishers, pp. 143–152, https://doi.org/10.11647/OBP.0300.08.
15. Nigam, Y. and M.R. Wilson, The Antimicrobial Activity of Medicinal Maggots, in A Complete Guide to Maggot Therapy: Clinical Practice, Therapeutic Principles, Production, Distribution, and Ethics, F. Stadler (ed.). 2022, Cambridge: Open Book Publishers, pp. 153–174, https://doi.org/10.11647/OBP.0300.09.
16. Nigam, Y. and M.R. Wilson, Maggot-assisted Wound Healing, in A Complete Guide to Maggot Therapy: Clinical Practice, Therapeutic Principles, Production, Distribution, and Ethics, F. Stadler (ed.). 2022, Cambridge: Open Book Publishers, pp. 175–194, https://doi.org/10.11647/OBP.0300.10.
17. Pianka, E.R., R-selection and K-selection. The American Naturalist, 1970. 104(940): p. 592–597, https://doi.org/10.1086/282697.
18. Rueda, L.C., et al., Lucilia sericata Strain from Colombia: Experimental Colonization, Life Tables and Evaluation of Two Artificial Diets of the Blowfly Lucilia sericata (Meigen) (Diptera: Calliphoridae), Bogota, Colombia Strain. Biological Research, 2010. 43(2): pp. 197–203, https://dx.doi.org/10.4067/S0716-97602010000200008.
19. Mackerras, M.J., Observations on the Life-histories, Nutritional Requirements and Fecundity of Blowflies. Bulletin of Entomological Research, 1933. 24: pp. 353–362, https://dx.doi.org/10.1017/S0007485300031680.
20. SolasCure. SolasCure. 2021. https://solascure.com/.
21. Close, B., et al., Recommendations for Euthanasia of Experimental Animals: Part 1. Laboratory Animals, 1996. 30(4): pp. 293–316, https://doi.org/10.1258/002367796780739871.
22. Cooper, J.E., Anesthesia, Analgesia, and Euthanasia of Invertebrates. ILAR Journal, 2011. 52(2): pp. 196–204, https://doi.org/10.1093/ilar.52.2.196.
23. Lewbart, G.A. and C. Mosley, Clinical Anesthesia and Analgesia in Invertebrates. Journal of Exotic Pet Medicine, 2012. 21(1): pp. 59–70, https://doi.org/10.1053/j.jepm.2011.11.007.
24. Wound Healing and Management Node Group, Recommended Practice: Wound Debridement — Larval Therapy. Wound Practice and Research, 2014. 22(1): pp. 48–49.
25. Richards, C.S. and M.H. Villet, Data Quality in Thermal Summation Development Models for Forensically Important Blowflies. Medical and Veterinary Entomology, 2009. 23(3): pp. 269–276, https://doi.org/10.1111/j.1365-2915.2009.00819.x.
26. Rabêlo, K.C.N., et al., Bionomics of Two Forensically Important Blowfly Species Chrysomya megacephala and Chrysomya putoria (Diptera: Calliphoridae) Reared on Four Types of Diet. Forensic Science International, 2011. 210(1–3): pp. 257–262, https://doi.org/10.1016/j.forsciint.2011.03.022.
27. SA Health. Fact Sheet. Coral Handling Safety Tips for Aquarium Owners. https://www.sahealth.sa.gov.au/wps/wcm/connect/public+content/sa+health+internet/resources/coral+handling+safety+tips+for+aquarium+owners.
28. UNEP, Compendium of Technologies for Treatment/Destruction of Healthcare Waste. 2012, United Nations Environment Programme Division of Technology, Industry and Economics International Environmental Technology Centre: Osaka, Japan. https://www.uncrd.or.jp/content/documents/04_Chandak-UNEP%20IETC.pdf.
29. Stadler, F. and P. Takáč, Medicinal Maggot Production, in A Complete Guide to Maggot Therapy: Clinical Practice, Therapeutic Principles, Production, Distribution, and Ethics, F. Stadler (ed.). 2022, Cambridge: Open Book Publishers, pp. 289–330, https://doi.org/10.11647/OBP.0300.14.
30. Thomas, K., Overview and Introduction to Advance Care Planning, in Advance Care Planning in End of Life Care. 2017, Oxford: Oxford University Press, pp. 1–18. http://doi.org/10.1093/oso/9780198802136.003.0001.
31. Benbow, M., Ethics and Wound Management. Journal of Community Nursing, 2006. 20(3): pp. 24, 26, 28.
32. Chadwick, P., et al., Appropriate Use of Larval Debridement Therapy in Diabetic Foot Management: Consensus Recommendations. Diabetic Foot Journal, 2015. 18(1): pp. 37–42.
33. Petherick, E.S., et al., Patient Acceptability of Larval Therapy for Leg Ulcer Treatment: A Randomised Survey to Inform the Sample Size Calculation of a Randomised Trial. BMC Medical Research Methodology, 2006. 6: 43, https://doi.org/10.1186/1471-2288-6-43.
34. NSW Health. Consent to Medical and Healthcare Treatment Manual. 2020. https://www.health.nsw.gov.au/policies/manuals/Pages/consent-manual.aspx.
35. Gillon, R., Medical Ethics: Four Principles Plus Attention to Scope. BMJ, 1994. 309(6948): pp. 184–188, https://doi.org/10.1136/bmj.309.6948.184.
36. Dau, A.A., S. Tloba, and M.A. Daw, Characterization of Wound Infections among Patients Injured during the 2011 Libyan Conflict. Eastern Mediterranean Health Journal = La Revue De Santé De La Méditerranée Orientale = Al-Majallah Al-Ṣiḥḥīyah Li-Sharq Al-Mutawassiṭ, 2013. 19(4): pp. 356–361, http://applications.emro.who.int/emhj/v19/04/EMHJ_2013_19_4_356_361.pdf.
37. Eardley, W.G.P., et al., Infection in Conflict Wounded. Philosophical Transactions. Biological Sciences, 2011. 366(1562): pp. 204–218, https://doi.org/10.1098/rstb.2010.0225.
38. Sen, C.S., Human Wounds and Its Burden: An Updated Compendium of Estimates. Advances in Wound Care, 2019. 8(2): pp. 39–48, https://doi.org/10.1089/wound.2019.0946.
39. Wuthisuthimethawee, P., et al., Wound Management in Disaster Settings. World Journal of Surgery, 2015. 39(4): pp. 842–853, https://doi.org/10.1007/s00268-014-2663-3.
40. Ali, S.M., et al., The Personal Cost of Diabetic Foot Disease in the Developing World — A Study from Pakistan. Diabetic Medicine 2008. 25(10): pp. 1231–1233, https://doi.org/10.1111/j.1464-5491.2008.02529.x.
41. Fleischmann, W., M. Grassberger, and R. Sherman, Maggot Therapy. A Handbook of Maggot-Assisted Wound Healing. 2004, Stuttgart: Georg Thieme Verlag.
42. Mirabzadeh, A., et al., Maggot Therapy for Wound Care in Iran: A Case Series of the First 28 Patients. Journal of Wound Care, 2017. 26(3): pp. 137–143, https://doi.org/10.12968/jowc.2017.26.3.137.
43. PIC/S, Guide to Good Manufacturing Practice for Medicinal Products — Annexes. 2017, Pharmaceutical Inspection Convention Pharmaceutical Inspection Co-operation Scheme. https://picscheme.org/en/publications?tri=gmp#zone.
44. Chan, Q.E., M.A. Hussain, and V. Milovic, Eating out of the Hand, Maggots — Friend or Foe? Journal of Plastic, Reconstructive and Aesthetic Surgery, 2012. 65(8): pp. 1116–1118, https://doi.org/10.1016/j.bjps.2012.01.014.
45. Stadler, F., R.Z. Shaban, and P. Tatham, Maggot Debridement Therapy in Disaster Medicine. Prehospital and Disaster Medicine, 2016. 31(1): pp. 79–84, https://doi.org/10.1017/S1049023X15005427.
46. Bazyar, J., et al., The Principles of Triage in Emergencies and Disasters: A Systematic Review. Prehospital and Disaster Medicine, 2020. 35(3): pp. 305–313, https://doi.org/10.1017/S1049023X20000291.
47. Whitaker, I.S., et al., Larval Therapy from Antiquity to the Present Day: Mechanisms of Action, Clinical Applications and Future Potential. Postgraduate Medical Journal, 2007. 83(980): pp. 409–413, https://doi.org/10.1136/pgmj.2006.055905.
48. Stadler, F., Supply Chain Management for Maggot Debridement Therapy in Compromised Healthcare Settings. 2018. Unpublished doctoral dissertation, Griffith University, Queensland, https://doi.org/10.25904/1912/3170.
49. Takáč, P., et al., and F. Stadler, Establishment of a Medicinal Maggot Production Facility and Treatment Programme in Kenya, in A Complete Guide to Maggot Therapy: Clinical Practice, Therapeutic Principles, Production, Distribution, and Ethics, F. Stadler (ed.). 2022, Cambridge: Open Book Publishers, pp. 289–330, https://doi.org/10.11647/OBP.0300.14.
50. Ghosh, B.N., Rich Doctors and Poor Patients: Market Failure and Health Care Systems in Developing Countries. Journal of Contemporary Asia, 2008. 38(2): pp. 259–276, https://doi.org/10.1080/00472330701546525.
51. Humphreys, I., P. Lehane, and Y. Nigam, Could Maggot Therapy Be Taught in Primary Schools? Journal of Biological Education, 2020: pp. 1–11, https://doi.org/10.1080/00219266.2020.1748686.
52. Čičková, H., et al., Growth and Survival of Bagged Lucilia sericata Maggots in Wounds of Patients Undergoing Maggot Debridement Therapy. Evidence-based Complementary and Alternative Medicine, 2013. https://doi.org/10.1155/2013/192149.
53. Stadler, F. and P. Tatham, Drone-assisted Medicinal Maggot Distribution in Compromised Healthcare Settings, in A Complete Guide to Maggot Therapy: Clinical Practice, Therapeutic Principles, Production, Distribution, and Ethics, F. Stadler (ed.). 2022, Cambridge: Open Book Publishers, pp. 383–402, https://doi.org/10.11647/OBP.0300.18.
54. MedMagLabs. MedMagLabs. http://medmaglabs.com.
55. Sherman, R.A. and M.R. Hetzler, Maggot Therapy for Wound Care in Austere Environments. Journal of Special Operations Medicine, 2017. 17(2): pp. 154–162.
56. Williams, J., R. World Medical Association Medical Ethics Manual. 2015. https://www.wma.net/wp-content/uploads/2016/11/Ethics_manual_3rd_Nov2015_en.pdf.
57. Braun, W., Jainism, in World Religions for Healthcare Professionals, S. Ssorajjakool, et al. (eds). 2017, Florence, United Kingdom: Taylor & Francis Group.
