3. Wound Aetiologies, Patient Characteristics, and Healthcare Settings Amenable to Maggot Therapy
© 2022 Sherman & Stadler, CC BY-NC 4.0 https://doi.org/10.11647/OBP.0300.03
It is important for healthcare practitioners to understand when to use maggot therapy. This chapter explains the general factors that determine the choice of wound treatment and how they apply to maggot therapy: i) the wound characteristics, ii) the patient characteristics, iii) the environment, iv) the available resources, and v) the specific characteristics of each available treatment modality. Beyond the regular healthcare setting, maggot therapy can make a significant contribution to the treatment of people with wounds in compromised healthcare settings such as in times of disaster and armed conflict, in underserved populations, or in palliative care.
Introduction
There is a great need for improved wound care in both resourced and compromised healthcare settings. Although not a panacea, maggot therapy can meet many of these pressing wound care needs. It is well-established that medicinal maggots have three major actions on wounds: 1) they dissolve and dislodge dead (necrotic) tissue and debris (debridement), 2) they kill microorganisms (“disinfection”), and 3) they stimulate healthy tissue to grow faster, as explained in Chapters 8 to 10 of this book [1–3]. Therefore, maggot therapy can usually assist in treating wounds that require any of these actions. Notwithstanding this broad spectrum of potential indications, it is important for healthcare policy makers, regulators, and health administrators to understand the contribution maggot therapy can make to wound care in order to support mainstreaming of this therapy [4]. It is also important for healthcare practitioners to understand when to use maggot therapy.
While treatment selection can seem quite complicated at times, the following general factors should always be considered when choosing a wound treatment: i) the wound characteristics, ii) the patient characteristics, iii) the environment, iv) the available resources, and v) the specific characteristics of each available treatment modality. Examples of how these factors might affect treatment decisions can be found in Table 3.1. Using these characteristics as a guiding framework, this chapter explains when maggot therapy is indicated.
It should also be noted that the specific types of wounds and situations that are appropriately treated with a particular method or product are usually based on scientific studies, anecdotal reports, personal experience, and regulatory approvals. Since maggot therapy is not officially regulated in most countries, this discussion of indications for maggot therapy is based primarily on its mechanisms of action and on published experience.
Table 3.1 Factors to consider when selecting wound treatments.
|
Factor |
Considerations |
|
i) Wound characteristics |
|
|
ii) Patient characteristics |
|
|
iii) Environmental characteristics |
|
|
iv) Resource availability |
|
|
v)Characteristics of the treatment modality |
|
Wounds Amenable to Maggot Therapy
Wound Characteristics
Necrosis. Most wounds on an otherwise healthy person will heal quite well despite our choice of therapy. But when dead tissue covers or fills that wound, then the surrounding healthy tissue cannot fill the void. What’s more, microorganisms that live and feed on dead tissue, or under its cover, can multiply and spread beyond these borders, especially if they can also live in the surrounding live tissue. Even if they cannot themselves spread beyond dead tissue, their secretions (called “toxins”) may be harmful to the surrounding live tissue, and may cause that tissue, too, to become inflamed and/or die, thereby extending the wound. Although scientific proof is sparse, there is wide agreement that a wound with necrotic tissue and debris will not easily heal until the necrotic tissue and debris is removed [5, 6].
Maggots must compete fiercely with other scavengers for limited resources, and many of those competing scavengers would consume the maggots, too, if the maggots were found still on the rotting tissue. The maggots used therapeutically are those that are well-adapted to quickly remove (and ingest) as much of the necrotic tissue and debris as possible, and then leave the area. Maggot therapy debrides the necrotic wound by at least two methods: enzymatic debridement and physical debridement [7]. The maggot’s digestive juices, rich with proteolytic enzymes, are excreted into the wound bed and quickly dissolve dead tissue. Meanwhile, as the maggot crawls about the wound bed, its microscopic spines and its modified mandibles (“mouth hooks”) physically dislodge some of the debris, helping the digestive juices gain access into the crevices. A more detailed discussion of the maggot’s debridement action can be found in Chapter 8 [1].
Infection. Given that medicinal blowflies, if living in the wild, would inhabit corpses, faeces, and other rotting organic matter, it seems logical that the larvae would have a method for killing infectious organisms, or else the microbes might kill the maggots. The mechanisms by which the maggots accomplish microbial killing are covered by Nigam and Wilson in Chapter 9 [2]. For the purpose of this discussion, let it suffice to acknowledge that medicinal maggots kill microbes through a variety of mechanisms including ingestion [8, 9], the secretion of antimicrobial compounds [10–15], dissolving biofilm [12, 16–21] and altering the local environment in ways that make it less hospitable to microbial pathogens [22, 23]. The end result is that microbial populations are reduced or eliminated [24–26] and clinical infections subside [27, 28]. The antimicrobial effect may even endure well beyond the life of the maggots [29].
Wound moisture content. Wounds can vary greatly in wetness. Some wounds are extremely dry; the necrotic tissue covering the wound being like leather (“eschar”). Other wounds may drain serous (watery), sero-sanguinous (bloody), or purulent (pus-filled) liquid profusely. The same wound may switch back and forth over the course of a week. Many dressings are suitable only for moderately dry wounds, or only for very wet wounds. Some dressings will not adhere to a wet wound; others are designed to absorb excess fluid tenaciously, making them superfluous or even dangerous for use with a minimally moist wound. Medicinal maggots have no such limitations, though the dressings used to confine them to the wound may themselves need to be selected or modified to meet the moisture conditions of the wound and surrounding tissue. Dressings will be discussed in detail in Chapter 5 [30].
Absence of healing—the chronic wound. Maggot therapy has been observed to enhance wound healing, even in apparently clean but stagnant wounds, at least as far back as William Baer’s time [31]. Subsequent clinicians have described, in controlled studies, the rapid proliferation of granulation tissue and hastened closing of the wound margins in previously stagnant, non-healing wounds [32–35]. The mechanisms known to be involved are described by Nigam and Wilson in Chapter 10 [3]. More and more therapists are using maggot therapy to promote healing and closure of chronic wounds, even though the wounds may not appear grossly necrotic nor infected. Some of the therapists describe this indication as maintenance debridement, others say their intent is growth-stimulation. The desired endpoint is the same: not just a clean (debrided) wound but a clean wound that is healing.
Wounds Treated with Maggot Therapy
Pressure ulcers/injuries. Pressure alone, from lying in the same position for a prolonged time, or in combination with shear force, can starve tissue of oxygenated blood which leads to tissue death. Shear may exacerbate the injury and cause mechanical damage to the compromised tissue [36]. These injuries lead to open necrotic ulcers that often fail to heal (Figure 3.1a). Maggot therapy is frequently used to treat pressure ulcers or injuries, and several controlled studies demonstrate its efficacy [33, 35, 37–39].
Arterial ulcers. Factors other than pressure can also prevent arterial blood from reaching tissue. Primary arterial disease, for example (i.e., vasculitis, arteritis obliterans, or thromboses) can occlude arteries and arterioles, leading to local necrosis of the skin and soft tissue. Many anecdotal reports support the use of maggot therapy for debriding these wounds [40–43]. However, without adequate blood flow, healing cannot occur even in the maggot-debrided wound [40, 41]. Therefore, the arterial flow needs to be assessed prior to maggot therapy. Even if arterial flow cannot be restored, maggot therapy can be administered to palliate ulcer symptoms such as exudate and odour and to prevent infection, thereby maintaining quality of life.
Venous stasis leg ulcers. Venous disease, too, can lead to skin and soft-tissue wounds. Venous insufficiency results in legs, for example, with increased intravascular pressure. The result is that fluid (serum) leaks out of the blood vessels and into the soft tissue, thereby causing swelling, pain, itching (trauma and infection from scratching). Oxygen cannot easily perfuse the fluid-filled (oedematous) leg, and the normal immune mechanisms (i.e., migration of white blood cells) are similarly impaired. In the end, skin breaks down more easily, the body does not fight the infections well, and healing is impaired (Figure 3.1b). Venous ulcers are best prevented by compression and elevation of the affected limb. However, when a person presents with a venous ulcer, then regular debridement is necessary along with compression and elevation of the limb. Multiple controlled and non-controlled maggot therapy studies have described faster debridement (with or without faster healing) of venous ulcers [38, 44–46].
Diabetic foot disease/diabetes related foot ulcers (DFUs). Diabetes results in multi-system disease. Neuropathy can lead to increased trauma, impaired immunity interferes with the body’s ability to fight infection, and blood vessel disease greatly inhibits healing (Figures 3.1c, 3.1d). DFUs require timely and expert assessment to identify the aetiology and prompt initiation of tailored management [47]. When aetiology is not ascertained, management will fail to address the underlying cause, which may lead to amputation [48]. Up to 85% of amputations can be prevented [49, 50]. Maggot therapy is often used to debride these wounds and control emerging infections. Several controlled studies [32, 34] and case series [51] have demonstrated significant benefits.
Traumatic and post-surgical wounds. Medicinal maggots are quite useful in debriding traumatic and infected or necrotic post-surgical wounds (Figure 3.1e, 3.1f). In an otherwise healthy individual, these wounds usually heal quite well after initial debridement and infection management.
Burns. Unlike many other necrotic wounds, burns need to be debrided acutely (Figure 3.1g). When the burn area is extensive or when vital structures could be damaged by surgical debridement, maggot debridement has proven to be a good alternative [10, 52–58]. Burn wounds are often painful, and maggot therapy is not likely to be less painful than any other manipulation of the damaged tissue. Therefore, anaesthetics will be just as necessary during maggot debridement as during any other form of debridement, and maybe more so.
Undermining wounds, difficult to reach or visualise. Because medicinal maggots, by nature, crawl into nooks and crannies, they are very appropriate for treating wounds whose boundaries may not be easily visualised. Like miners, the larvae will crawl into the depths of undermined tissue and sinus tracts, debriding within these areas until they need to return to the surface for air, food (assuming that the area no longer contains necrotic tissue or nutritious liquid), or to moult. Treating undermining wounds with maggot therapy can avoid having to open a deep cavity for accurate visualisation and debridement.
Body cavities. Similar to undermined wounds and sinus tracts, medicinal maggots have been used effectively to debride body cavities with infected necrotic tissue, such as necrotising peritonitis or chronic empyema [56]. If debridement is not needed in the crevices or out-of-sight areas of these cavities, it may be possible and preferable to apply the maggots within sealed mesh bags so that they stay contained and can be more easily removed, as explained in Chapter 5 [30].
Bones and Joints. Bone infections (“osteomyelitis”) were a common indication for maggot therapy during the 1930s, when many orthopaedic surgeons used maggot therapy to perform the “fine debridement” after the surgeon removed the grossly infected or dead bone [31, 59, 60]. We now have procedures and antibiotics that make post-operative infections much less common. Maggot therapy is rarely used for osteomyelitis at all anymore; osteomyelitis is generally considered a surgical disease, treated by removing (resecting) the dead bone and by long-term antibiotics to prevent spread or recurrence of the infection. However, when surgical resection and/or antibiotics are inadequate or are not an option, maggot therapy is sometimes used to debride the bone [61–63]. Similarly, infected joints and joint prostheses are generally treated by removing the infected hardware and draining the infection, along with antibiotic coverage. But when the removal of hardware is risky or impossible, maggot therapy has been used alongside the antibiotics to eliminate the infection [64, 65].
Malignancy. Medicinal maggots dissolve necrotic tissue, but not viable tissue. In that regard, they are not useful for killing live cancer cells. However, when a tumour mass outstrips its blood supply and starts to rot, maggot therapy can be a rapid and effective method for removing the necrotic mass (Figure 3.1h). Many cancer patients were able to return to a reasonably normal life in public only after maggot therapy removed their foul-smelling, draining, painfully infected tumours [66–68].
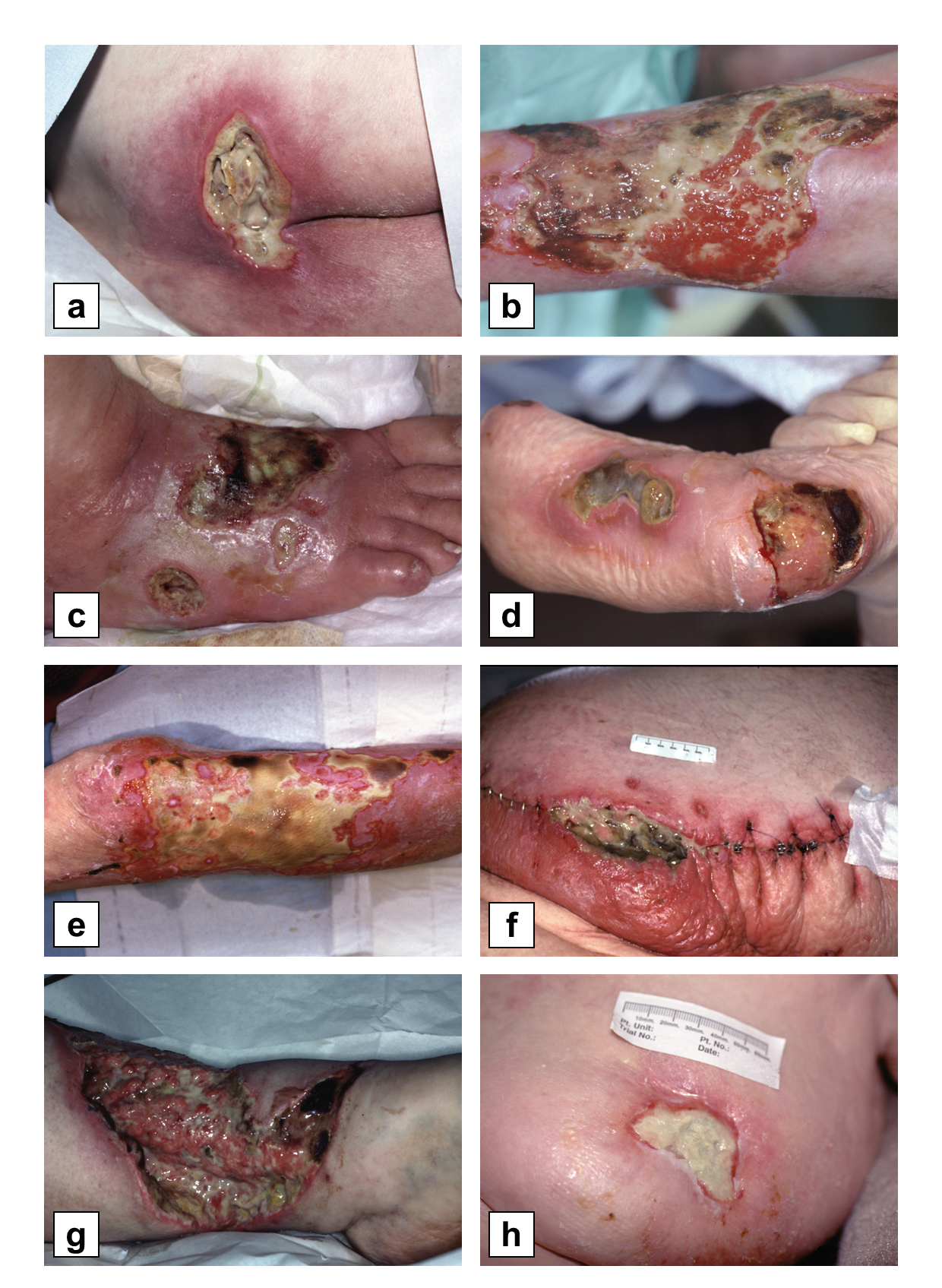
Figure 3.1 Chronic wounds amenable to maggot therapy: a) pressure ulcer, b) venous stasis ulcer, c&d) diabetic foot ulcers, e) burn, f) surgical wound dehiscence, g) orthopaedic wound, h) malignancy. Photos: © Steve Thomas, www.medetec.co.uk.
Specific Infections
Multi-resistant bacteria. Because blowflies kill microbes through mechanisms other than those used by typical antibiotics [2], they are effective in killing pathogens that have developed resistance to those antibiotics, such as methicillin-resistant Staphylococcus aureus (MRSA) [66–68]; and multi-resistant Acinetobacter [69].
Fungal infections. Maggot secretions kill or inhibit representative fungi in the laboratory [70, 71], though very little clinical experience with fungi has been published [72].
Fasciitis. Several microbes, and especially mixed aerobic and anaerobic infections, can be highly destructive to soft tissue, causing necrosis that rapidly extends down to the deep layers of muscle and bone. Fasciitis is a surgical emergency. Even with immediate resection of the dead tissue, systemic broad-spectrum antibiotics and intensive care, the mortality of these infections can still exceed 40% [73]. Frequently, repeated surgical debridement is required, ultimately leading to destruction or resection of vital structures, and the need for major anatomical reconstruction, if the patient survives at all. Maggot therapy has been used successfully to debride these wounds without unnecessarily harming the remaining viable tissue [74–77]. In a small but prospective study, patients with Fournier’s gangrene that were debrided only by maggots if they required more than one initial surgical resection, not only survived their wounds without the need for any additional surgical debridement but also avoided the need for reconstructive surgery [78].
Tropical ulcers (Mycobacteriosis and tropical phagedenic ulcers). The term “tropical ulcers” collectively refers to painful wounds in typically malnourished individuals living in tropical climates. The wounds often follow trauma, and they may be colonised, if not caused, by Mycobacterium ulcerans (Buruli ulcer, mycobacteriosis) or Fusobacterium and Borrelia (Phagedenic ulcers, Naga sores) [79]. These are chronic wounds often with very poor outcomes, though antibiotics and improved nutrition can be curative. Buruli ulcers (Mycobacterium ulcerans wounds) often deteriorate following what appears to be appropriate antimicrobial therapy [80]. Medicinal maggots were used extensively in the 1930s as an adjunct to surgical resection, in the treatment of tuberculous osteomyelitis. It is reasonable to assume that maggot therapy should adequately debride tropical ulcer wounds as well, but the efficacy and safety of maggot debridement for these wounds remain undefined.
The wounds commonly seen in patients living with leprosy (caused by Mycobacterium leprae) are generally caused by trauma; the leprosy bacterium is not necessarily within the wound [81]. These wounds should also respond to maggot therapy, if needed, just like any other traumatic wound.
Protozoans. Medicinal maggots have demonstrated anti-leishmania activity in vitro and in animal models [82–85], though no human studies have been reported. The endemic trepanematoses (i.e., yaws, pinta, and bejel) can also cause problematic soft-tissue wounds, but penicillin and several other readily available antibiotics are generally curative. No experience treating these lesions with maggot therapy has been published to date.
Population and Patient Characteristics
Population Characteristics
Disaster and conflict casualties. During a disaster or military conflict, the number of victims often overwhelms the capacity of first responders. This is the perfect opportunity to recruit fly larvae to assist in wound care [86]. Often there are insufficient facilities or hospital beds, and unreliable electricity and water supplies. Again, maggot therapy is well-suited to these trying circumstances because the larvae require none of those amenities. However, the supply chains for getting medicinal maggots to compromised healthcare settings are hitherto non-existent. Because maggots are highly perishable and cannot be stored for more than a day or two, attention needs to be paid to their production close to the point of care in disasters and conflict, or to reliable logistics solutions that ensure timely and safe delivery of medicinal maggots [87, 88].
Rural, tropical, and medically underserved populations. People living in sparsely populated or medically underserved regions have all the same problems as described above, but often in different proportions. Wounds as a result of infectious and parasitic diseases, burns, and traumatic injury are common, as are increasingly wounds related to cancer, diabetes and cardiovascular disease. Again, maggot therapy is a great resource when there is a severe shortage of medical staff and other healthcare resources, provided that reliable supply chains exist to make maggot therapy accessible to these communities.
End of life and palliative care residents. Death is a part of life whether in a rural part of the world or in a big city. Most people become ill or reach an older age before dying. With aging and chronic illness, organs (including skin) begin to fail. Skin failure is a common pre-terminal occurrence [89]. As already noted, maggot therapy is a simple and effective means of treating necrotic, infected, or chronic wounds in the ill or frail person [90]. Maggot therapy can quickly provide surgical-quality wound care, at home or in long-term/residential care, at a fraction of the cost of a surgeon, and be cost-effective when compared to existing advanced dressings that deliver only limited wound bed improvement.
Characteristics of Persons with Wounds
Age and gender. Many treatment modalities are not available to persons with wounds depending on their particular age, gender, or both. For example, certain antibiotics and other pharmaceuticals should not be used in children or in child-bearing women, or even in women of a particular (“child-bearing”) age. Often this is the result of real or theoretical complications that have been observed in humans or in animal models. Other times it is simply because the studies supporting safety and efficacy were performed in populations that excluded these high-risk individuals. Maggot therapy does not need to be withheld from anyone on the basis of gender or age [31, 91].
Frailty. Whether due to age or underlying medical illnesses, some people are unable to endure the physical or mental stress of aggressive medical interventions such as surgery, anaesthesia, or even frequent phlebotomy (needles). Even if their cardiopulmonary status is stable, an underlying problem such as hypertension, obesity, emphysema or sub-optimally managed diabetes may make surgery and/or anaesthesia excessively risky [92]. These conditions are not contraindications to maggot therapy, because maggot therapy does not require general anaesthesia, and does not put any additional physical stress on the body.
Prognosis. Non-healing wounds are often seen in people when they are dying. When managing wounds that cannot heal, including in people who are dying, the focus of holistic management is to optimise quality of life and to maintain optimal dignity. Maggot therapy can be of particular benefit if wounds are infected, foul-smelling, draining, painful, in need of much care and attention. Maggot therapy controls the infection, debrides the necrotic tissue, and thereby diminishes or eliminates the drainage, odour and pain of a necrotic wound or terminal ulcer. Maggot therapy can provide comfort in these ways, without adding additional stress to a patient in their last days of life [77].
Need for other medical interventions. Maggot therapy can be used safely and effectively even in people receiving other drugs and treatments. Medicinal maggots and their debridement efficacy appear to be unaffected by antibiotics [93, 94] and most other drugs in pharmacologic doses, and various medical and life support interventions [56, 95–97]. In fact, maggots have proven to be safe and effective in patients receiving anticoagulants or intensive care that made surgical debridement unsafe [56, 98]. Unpublished anecdotal reports suggest that medicinal maggots may not survive on animals receiving systemic or topical insecticides, such as ivermectin or flea and tick treatments. No work has been published concerning the impact of cancer chemotherapy on medicinal maggots.
Environmental Characteristics
One of the most favourable aspects of maggot therapy is its flexibility regarding the care setting. Patients can be treated as in-patients, as outpatients, in the community, in field hospital situations, and even in the most austere healthcare settings that lack any medical resources [31, 86, 99]. This is because the application of, and treatment with, medicinal maggot dressings does not require energy or sophisticated equipment. Maggots are also well-adapted to unhygienic environments and therefore will perform their therapeutic action equally well in the modern hospital environment, the battlefield, and the disaster zone. The lack of stable shelter is not an impediment to using maggot therapy [86, 99]. Maggot therapy will still be effective, even if exposed to the elements, as long as the maggots receive enough air and the wounds are well-drained to prevent suffocation and drowning. Extreme climatic conditions should not be a deterrent either. In general, if the weather is tolerable to the patient, it will be tolerated by medicinal maggots [100, 101]. Wound temperatures encountered by medicinal maggots will depend on the body region, general condition of the patient and the environmental temperature, but will in most instances not be lower than 30℃ or exceed 40℃, which is a temperature range that promotes high activity and rapid growth in medicinal maggots [100, 102] and lies well below the lethal temperature threshold of around 47℃ [103]. Environmental temperature is only a concern during transport of young medicinal maggots that are deprived of food until they reach the point of care and patient. To slow down metabolic activity and maintain optimal health, medicinal maggots should be kept at 6 to 25℃ and be applied to the wound within 48 hours of dispatch [87]. In other words, most limitations imposed on healthcare provision by the environment, facilities, or equipment will have little to no impact on the work of the maggots.
Resources
Social resources. Social support systems are occasionally called into play to help with medical issues. In some parts of the world, patients are discharged to home while still recovering. Friends and family may need to assist in the care of their loved ones at home or in the hospital. Maggot therapy is simple enough that dressings can be applied, maintained, and/or removed by the patient or the patient’s caregivers [104].
Financial resources. Without financial resources, the purchase of specialised medical supplies can seem like an insurmountable problem. Fortunately, medicinal maggots are not very costly to produce (except when regulatory agencies are involved). Many labs provide free or subsidised larvae to patients with limited finances. Some therapists procure larvae themselves when a commercial product is unavailable [99, 105]. Even when financial resources are sparse, maggot therapy remains a viable and cost-effective solution for wound care.
Moreover, the use and application of medicinal maggots requires no electricity, no batteries, no running or sterile water, and no refrigeration [86, 99]. Maggot dressings can be made from materials readily available at any facility that is modestly prepared to manage wounds (e.g. gauze and tape). While specialised or maggot-specific dressing supplies can simplify the application of maggot therapy, such amenities are not required. Please refer to Chapter 5 [30] for a discussion of dressing options.
Summary
While maggot therapy is not a panacea and will not resolve the underlying causes, particularly of chronic wounds, it is appropriate for the treatment of a wide variety of infected, necrotic, non-healing skin and soft-tissue wounds. Maggot therapy may also be used for acute wounds in need of urgent debridement, and non-necrotic wounds that simply will not close. In all of these situations, maggot therapy has proven to be very effective and relatively safe, at a low or reasonable cost. Maggot therapy can make a significant contribution to the treatment of people with wounds in disasters and armed conflict, in underserved populations, and in palliative care. With respect to patient characteristics, maggot therapy is also highly accommodating as it can be used to treat people of all ages, genders and levels of frailty. Indeed, it should be a treatment of choice for debridement and wound maintenance where people are unfit to endure general anaesthesia or are at their end of life. Moreover, medicinal maggots can be deployed in both high- and low-resource care settings irrespective of healthcare infrastructure and hygiene levels as they do not require any special facilities, resources, or highly-skilled personnel. Table 3.2 provides a summary of the ideal wound dressing characteristics, and how maggot therapy compares favourably to that ideal.
Table 3.2 The degree to which maggot therapy compares with the ideal wound dressing.
|
How maggot therapy compares |
|
|
Effective for many wound types |
***** |
|
Safe for healthy wound tissue |
***** |
|
Can be applied by non-healthcare professionals |
***** |
|
Can be applied with minimal training |
***** |
|
Low environmental impact |
***** |
|
Low cost |
***** |
|
Safe for healthy surrounding tissue |
**** |
|
Low maintenance |
**** |
|
Simple to discard |
**** |
|
Patient/cultural acceptability |
**** |
|
Healthcare provider acceptability |
* |
|
Easy availability/convenience |
* |
References
1. Nigam, Y. and M.R. Wilson, Maggot Debridement, in A Complete Guide to Maggot Therapy: Clinical Practice, Therapeutic Principles, Production, Distribution, and Ethics, F. Stadler (ed.). 2022, Cambridge: Open Book Publishers, pp. 143–152, https://doi.org/10.11647/OBP.0300.08.
2. Nigam, Y. and M.R. Wilson, The Antimicrobial Activity of Medicinal Maggots, in A Complete Guide to Maggot Therapy: Clinical Practice, Therapeutic Principles, Production, Distribution, and Ethics, F. Stadler (ed.). 2022, Cambridge: Open Book Publishers, pp. 153–174, https://doi.org/10.11647/OBP.0300.09.
3. Nigam, Y. and M.R. Wilson, Maggot-assisted Wound Healing, in A Complete Guide to Maggot Therapy: Clinical Practice, Therapeutic Principles, Production, Distribution, and Ethics, F. Stadler (ed.). 2022, Cambridge: Open Book Publishers, pp. 175–194, https://doi.org/10.11647/OBP.0300.10.
4. Bullen, B. and P. Chadwick, Clinical Integration of Maggot Therapy, in A Complete Guide to Maggot Therapy: Clinical Practice, Therapeutic Principles, Production, Distribution, and Ethics, F. Stadler (ed.). 2022, Cambridge: Open Book Publishers, pp. 97–118, https://doi.org/10.11647/OBP.0300.06.
5. Falabella, A.F., Debridement and Wound Bed Preparation. Dermatologic Therapy, 2006. 19(6): pp. 317–325, https://doi.org/10.1111/j.1529-8019.2006.00090.x.
6. Weir, D., P. Scarborough, and J. Niezgoda, Wound Debridement, in Chronic Wound Care: The Essentials, D.L. Krasner and L. van Rijswijk (eds). 2018, HMP Communications: Malvern, PA, pp. 63–78.
7. Sherman, R.A., Mechanisms of Maggot-Induced Wound Healing: What Do We Know, and Where Do We Go from Here? Evidence-based Complementary and Alternative Medicine, 2014. 2014: pp. 1–13, https://doi.org/10.1155/2014/592419.
8. Greenberg, B., Model for Destruction of Bacteria in the Midgut of Blow Fly Maggots. Journal of Medical Entomology, 1968. 5(1): pp. 31–38, https://doi.org/10.1093/jmedent/5.1.31.
9. Mumcuoglu, K.Y., et al., Destruction of Bacteria in the Digestive Tract of the Maggot of Lucilia sericata (Diptera: Calliphoridae). Journal of Medical Entomology, 2001. 38(2): pp. 161–166, https://doi.org/10.1603/0022-2585-38.2.161.
10. Bian, H., et al., Beneficial Effects of Extracts from Lucilia sericata Maggots on Burn Wounds in Rats. Molecular Medicine Reports, 2017. 16(5): pp. 7213–7220, https://doi.org/10.3892/mmr.2017.7566.
11. Erdmann, G.R. and S.K. Khalil, Isolation and Identification of Two Antibacterial Agents Produced by a Strain of Proteus Mirabilis Isolated from Larvae of the Screwworm (Cochliomyia hominivorax) (Diptera: Calliphoridae). Journal of Medical Entomology, 1986. 23(2): pp. 208–211, https://doi.org/10.1093/jmedent/23.2.208.
12. Gordya, N., et al., Natural Antimicrobial Peptide Complexes in the Fighting of Antibiotic Resistant Biofilms: Calliphora Vicina Medicinal Maggots. PloS ONE, 2017. 12(3): e0173559, https://doi.org/10.1371/journal.pone.0173559.
13. Pöppel, A.-K., et al., Antimicrobial Peptides Expressed in Medicinal Maggots of the Blow Fly Lucilia sericata Show Combinatorial Activity against Bacteria. Antimicrobial Agents and Chemotherapy, 2015. 59(5): pp. 2508–2514, https://doi.org/10.1128/AAC.05180-14.
14. Teh, C.H., et al., Determination of Antibacterial Activity and Minimum Inhibitory Concentration of Larval Extract of Fly via Resazurin-based Turbidometric Assay. BMC Microbiology, 2017. 17(1): pp. 36–36, https://doi.org/10.1186/s12866-017-0936-3.
15. Wright, E.A. and E.R. Pavillard, An Antibiotic from Maggots. Nature, 1957. 180(4592): pp. 916–917, https://doi.org/10.1038/180916b0.
16. Bohova, J., et al., Selective Antibiofilm Effects of Lucilia sericata Larvae Secretions/Excretions against Wound Pathogens. Evidence-Based Complementary and Alternative Medicine, 2014. 2014: pp. 857360–857360, https://doi.org/10.1155/2014/857360.
17. Brown, A., et al., Blow Fly Lucilia sericata Nuclease Digests DNA Associated with Wound Slough/Eschar and with Pseudomonas aeruginosa Biofilm. Medical and Veterinary Entomology, 2012. 26(4): pp. 432–439, https://doi.org/10.1111/j.1365-2915.2012.01029.x.
18. Cazander, G., et al., Maggot Excretions Inhibit Biofilm Formation on Biomaterials. Clinical Orthopaedics and Related Research, 2010. 468(10): pp. 2789–2796, https://doi.org/10.1007/s11999-010-1309-5.
19. Cazander, G., et al., The Influence of Maggot Excretions on PAO1 Biofilm Formation on Different Biomaterials. Clinical Orthopaedics and Related Research, 2009. 467(2): pp. 536–545, https://doi.org/10.1007/s11999-008-0555-2.
20. Harris, L.G., et al., Disruption of Staphylococcus Epidermidis Biofilms by Medicinal Maggot Lucilia sericata Excretions/Secretions. International Journal of Artificial Organs, 2009. 32(9): pp. 555–564, https://doi.org/10.1177/039139880903200904.
21. van der Plas, M.J.A., et al., Maggot Excretions/Secretions Are Differentially Effective against Biofilms of Staphylococcus aureus and Pseudomonas aeruginosa. Journal of Antimicrobial Chemotherapy, 2008. 61(1): pp. 117–122, https://doi.org/10.1093/jac/dkm407.
22. Davydov, L., Maggot Therapy in Wound Management in Modern Era and a Review of Published Literature. 2011, SAGE Publications: Los Angeles, CA, pp. 89–93.
23. Nigam, Y., et al., Maggot Therapy: The Science and Implication for CAM Part II — Maggots Combat Infection. Evidence-based Complementary and Alternative Medicine, 2006. 3(3): pp. 303–308, https://doi.org/10.1093/ecam/nel022.
24. Contreras-Ruiz, J., et al., [Comparative Study of the Efficacy of Larva Therapy for Debridement and Control of Bacterial Burden Compared to Surgical Debridement and Topical Application of an Antimicrobial]. Gaceta médica de México, 2016. 152(Suppl 2): pp. 78–87, http://www.anmm.org.mx/GMM/2016/s2/GMM_152_2016_S2_78-87.pdf.
25. Malekian, A., et al., Efficacy of Maggot Therapy on Staphylococcus aureus and Pseudomonas aeruginosa in Diabetic Foot Ulcers: A Randomized Controlled Trial. Journal of Wound, Ostomy and Continence Nursing, 2019. 46(1): pp. 25–29, https://doi.org/10.1097/WON.0000000000000496.
26. Tantawi, T.I., et al., Clinical and Microbiological Efficacy of MDT in the Treatment of Diabetic Foot Ulcers. Journal of Wound Care, 2007. 16(9): pp. 379–383, https://doi.org/10.12968/jowc.2007.16.9.27868.
27. Armstrong, D.G., et al., Maggot Therapy in “Lower-extremity Hospice” Wound Care: Fewer Amputations and More Antibiotic-free Days. Journal of the American Podiatric Medical Association, 2005. 95(3): pp. 254–257, https://doi.org/10.7547/0950254.
28. Steenvoorde, P. and J. Oskam, Use of Larval Therapy to Combat Infection after Breast-conserving Surgery. Journal of Wound Care, 2005. 14(5): pp. 212–213, https://doi.org/10.12968/jowc.2005.14.5.26778.
29. Sherman, R.A. and K.J. Shimoda, Presurgical Maggot Debridement of Soft Tissue Wounds Is Associated with Decreased Rates of Postoperative Infection. Clinical Infectious Diseases, 2004. 39(7): pp. 1067–1070, https://doi.org/10.1086/423806.
30. Sherman, R., Medicinal Maggot Application and Maggot Therapy Dressing Technology, in A Complete Guide to Maggot Therapy: Clinical Practice, Therapeutic Principles, Production, Distribution, and Ethics, F. Stadler (ed.). 2022, Cambridge: Open Book Publishers, pp. 79–96, https://doi.org/10.11647/OBP.0300.05.
31. Baer, W.S., The Treatment of Chronic Osteomyelitis with the Maggot (Larva of the Blow Fly). The Journal of Bone and Joint Surgery. American Volume, 1931. 13: pp. 438–475, https://doi.org/10.1007/s11999-010-1416-3.
32. Markevich, Y.O., et al., Maggot Therapy for Diabetic Neuropathic Foot Wounds. Diabetologia, 2000. 43: p. Suppl 1: A15.
33. Sherman, R.A., Maggot versus Conservative Debridement Therapy for the Treatment of Pressure Ulcers. Wound Repair and Regeneration, 2002. 10(4): pp. 208–214, https://doi.org/10.1046/j.1524-475X.2002.10403.x.
34. Sherman, R.A., Maggot Therapy for Treating Diabetic Foot Ulcers Unresponsive to Conventional Therapy. Diabetes Care, 2003. 26(2): pp. 446–451, https://doi.org/10.2337/diacare.26.2.446.
35. Sherman, R.A., F. Wyle, and M. Vulpe, Maggot Therapy for Treating Pressure Ulcers in Spinal Cord Injury Patients. The Journal of Spinal Cord Medicine, 1995. 18(2): pp. 71–74, https://doi.org/10.1080/10790268.1995.11719382.
36. European Pressure Ulcer Advisory Panel. National Pressure Injury Advisory Panel and Pan Pacific Pressure Injury Alliance. Prevention and Treatment of Pressure Ulcers/Injuries: Clinical Practice Guideline. The International Guideline. 2019. www.internationalguideline.com/static/pdfs/Quick_Reference_Guide-10Mar2019.pdf.
37. Gilead, L., K.Y. Mumcuoglu, and A. Ingber, The Use of Maggot Debridement Therapy in the Treatment of Chronic Wounds in Hospitalised and Ambulatory Patients. Journal of Wound Care, 2012. 21(2): pp. 78–85, https://doi.org/10.12968/jowc.2012.21.2.78.
38. Mumcuoglu, K.Y., et al., Maggot Therapy for the Treatment of Intractable Wounds: Maggot Therapy for Intractable Wounds Pharmacology and Therapeutics. International Journal of Dermatology, 1999. 38(8): pp. 623–627, https://doi.org/10.1046/j.1365-4362.1999.00770.x.
39. Polat, E., et al., Treatment of Pressure Ulcers with Larvae of Lucilia sericata. Türkiye Fiziksel Tıp ve Rehabilitasyon Dergisi, 2017. 63(4): pp. 307–312, https://doi.org/10.5606/tftrd.2017.851.
40. Fleischmann, W., et al., Biosurgery — Maggots, Are They Really the Better Surgeons? Chirurg, 1999. 70(11): pp. 1340–1346, https://doi.org/10.1007/s001040050790.
41. Igari, K., et al., Maggot Debridement Therapy for Peripheral Arterial Disease. Annals of Vascular Diseases, 2013. 6(2): pp. 145–149, https://doi.org/10.3400/avd.oa.13-00036.
42. Nishijima, A., et al., Effective Wound Bed Preparation Using Maggot Debridement Therapy for Patients with Critical Limb Ischaemia. Journal of Wound Care, 2017. 26(8): pp. 483–489, https://doi.org/10.12968/jowc.2017.26.8.483.
43. Rafter, L., Larval Therapy Applied to a Large Arterial Ulcer: An Effective Outcome. British Journal of Nursing, 2013. 22(Sup4): pp. S24-S30, https://doi.org/10.12968/bjon.2013.22.Sup4.S24.
44. Dumville, J.C., et al., Larval Therapy for Leg Ulcers (VenUS II): Randomised Controlled Trial. BMJ, 2009. 338(7702): pp. 1047–1050, https://doi.org/10.1136/bmj.b773.
45. Mudge, E., et al., A Randomized Controlled Trial of Larval Therapy for the Debridement of Leg Ulcers: Results of a Multicenter, Randomized, Controlled, Open, Observer Blind, Parallel Group Study. Wound Repair and Regeneration, 2014. 22(1): pp. 43–51, https://doi.org/10.1111/wrr.12127.
46. Wayman, J., et al., The Cost Effectiveness of Larval Therapy in Venous Ulcers. Journal of Tissue Viability, 2000. 10(3): pp. 91–94, https://doi.org/10.1016/S0965-206X(00)80036-4.
47. Schaper N, et al. IWGDF Guidelines on the Prevention and Management of Diabetic Foot Disease. 2019. https://iwgdfguidelines.org/wp-content/uploads/2019/05/IWGDF-Guidelines-2019.pdf.
48. Hingorani, A.M.D., et al., The Management of Diabetic Foot: A Clinical Practice Guideline by the Society for Vascular Surgery in Collaboration with the American Podiatric Medical Association and the Society for Vascular Medicine. Journal of Vascular Surgery, 2016. 63(2): pp. 3S-21S, https://doi.org/10.1016/j.jvs.2015.10.003.
49. International Diabetes Federation. IDF Diabetes Atlas, 7th Edition. 2015. https://www.idf.org/component/attachments/attachments.html?id=1093&task=download.
50. Krishnan, S., et al., Reduction in Diabetic Amputations over 11 Years in a Defined U.K. Population: Benefits of Multidisciplinary Team Work and Continuous Prospective Audit. Diabetes Care, 2008. 31(1): pp. 99–101, https://doi.org/10.2337/dc07-1178.
51. Marineau, M.L., et al., Maggot Debridement Therapy in the Treatment of Complex Diabetic Wounds. Hawaii Medical Journal, 2011. 70(6): pp. 121–124, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3233395/.
52. Jun-cheng, W., et al., Maggot Therapy for Repairing Serious Infective Wound in a Severely Burned Patient. Chinese Journal of Traumatology, 2012. 15(2): pp. 124–125, https://doi.org/10.3760/cma.j.issn.1008-1275.2012.02.012.
53. Feng, X., et al., Evaluation of the Burn Healing Properties of Oil Extraction from Housefly Larva in Mice. Journal of Ethnopharmacology, 2010. 130(3): pp. 586–592, https://doi.org/10.1016/j.jep.2010.05.044.
54. Li, Q.M.D., et al., Maggots of Musca Domestica in Treatment of Acute Intractable Wound. Surgery, 2009. 145(1): pp. 122–123, https://doi.org/10.1016/j.surg.2008.08.016.
55. Namias, N., et al., Biodebridement: A Case Report of Maggot Therapy for Limb Salvage after Fourth-degree Burns. Journal of Burn Care & Rehabilitation, 2000. 21(3): pp. 254–257, https://academic.oup.com/jbcr/article-abstract/21/3/254/4758239?redirectedFrom=fulltext.
56. Sherman, R.A., C.E. Shapiro, and R.M. Yang, Maggot Therapy for Problematic Wounds: Uncommon and Off-label Applications. Advances in Skin & Wound Care, 2007. 20(11): pp. 602–610, https://doi.org/10.1097/01.ASW.0000284943.70825.a8.
57. Summers, J.B. and J. Kaminski, Maggot Debridement Therapy (MDT) for Burn Wounds. Burns: Journal of the International Society for Burn Injuries, 2003. 29(5): pp. 501–502, https://doi.org/10.1016/s0305-4179(03)00059-7.
58. Vistnes, L.M., R. Lee, and G.A. Ksander, Proteolytic Activity of Blowfly Larvae Secretions in Experimental Burns. Surgery, 1981. 90(5): pp. 835–841.
59. McKeever, D.C., Maggots in Treatment of Osteomyelitis: A Simple Inexpensive Method. The Journal of Bone and Joint Surgery, 1933. 15: pp. 85–93, http://dx.doi.org/10.1007/s11999-008-0240-5.
60. Stewart, M.A., The Therapeutic Behavior of Lucilia Sericata Meig. Larvae in Osteomyelitis Wounds. Science (American Association for the Advancement of Science), 1934. 79(2055): pp. 459–460
61. Galeano, M., et al., Maggot Therapy for Treatment of Osteomyelitis and Deep Wounds: An Old Remedy for an Actual Problem [8]. Plastic and Reconstructive Surgery (1963), 2001. 108(7): pp. 2178–2179.
62. Horn, K.L., A.H. Cobb, and G.A. Gates, Maggot Therapy for Subacute Mastoiditis. Archives of otolaryngology (1960), 1976. 102(6): pp. 377–379, https://doi.org/10.1001/archotol.1976.00780110089013.
63. Mumcuoglu, K.Y., et al., [Maggot Therapy for Gangrene and Osteomyelitis]. Harefuah, 1997. 132(5): pp. 323–325, 382.
64. Townley, W.A., A. Jain, and C. Healy, Maggot Debridement Therapy to Avoid Prosthesis Removal in an Infected Total Knee Arthroplasty. Journal of Wound Care, 2006. 15(2): pp. 78–79, https://doi.org/10.12968/jowc.2006.15.2.26890.
65. Wollina, U., M. Kinscher, and H. Fengler, Maggot Therapy in the Treatment of Wounds of Exposed Knee Prostheses. International Journal of Dermatology, 2005. 44(10): pp. 884–886, https://doi.org/10.1111/j.1365-4632.2005.02366c.x.
66. Beasley, W.D. and G. Hirst, Making a Meal of MRSA—The Role of Biosurgery in Hospital-acquired Infection. 2004, Elsevier Ltd: London, pp. 6–9.
67. Bowling, F.L., E.V. Salgami, and A.J.M. Boulton, Larval Therapy: A Novel Treatment in Eliminating Methicillin-Resistant Staphylococcus aureus from Diabetic Foot Ulcers. Diabetes Care, 2007. 30(2): pp. 370–371, https://doi.org/10.2337/dc06-2348.
68. Dissemond, J., et al., Treatment of Methicillin-resistant Staphylococcus aureus (MRSA) as Part of Biosurgical Management of a Chronic Leg Ulcer. Hautarzt, 2002. 53(9): pp. 608–612, https://doi.org/10.1007/s00105-002-0336-x.
69. Čeřovský, V. and R. Bém, Lucifensins, the Insect Defensins of Biomedical Importance: The Story behind Maggot Therapy. Pharmaceuticals, 2014. 7(3): pp. 251–264, https://doi.org/10.3390/ph7030251.
70. Evans, R., E. Dudley, and Y. Nigam, Detection and Partial Characterization of Antifungal Bioactivity from the Secretions of the Medicinal Maggot, Lucilia sericata. Wound Repair and Regeneration, 2015. 23(3): pp. 361–368, https://doi.org/10.1111/wrr.12287.
71. Margolin, L. and P. Gialanella, Assessment of the Antimicrobial Properties of Maggots. International Wound Journal, 2010. 7(3): pp. 202–204, https://doi.org/10.1111/j.1742-481X.2010.00234.x.
72. Bohac, M., et al., Maggot Therapy in Treatment of a Complex Hand Injury Complicated by Mycotic Infection. Bratislava Medical Journal, 2015. 116(11): pp. 671–673, https://doi.org/10.4149/bll_2015_128.
73. Sorensen, M.D. and J.N. Krieger, Fournier’s Gangrene: Epidemiology and Outcomes in the General US Population. Urologia Internationalis, 2016. 97(3): pp. 249–259, https://doi.org/10.1159/000445695.
74. Angel, K., et al., Madentherapie bei Fournierscher Gangrän — erste Erfahrungen mit einer neuen Therapie. Aktuelle Urologie, 2000. 31(07): pp. 440–443.
75. Dunn, C., U. Raghavan, and A.G. Pfleiderer, The Use of Maggots in Head and Neck Necrotizing Fasciitis. The Journal of Laryngology & Otology, 2002. 116(1): pp. 70–72, https://doi.org/10.1258/0022215021910212.
76. Preuss, S.F., M.J. Stenzel, and A. Esriti, The Successful Use of Maggots in Necrotizing Fasciitis of the Neck: A Case Report. Head & Neck, 2004. 26(8): pp. 747–750, https://doi.org/10.1002/hed.20092.
77. Steenvoorde, P., et al., Maggot Debridement Therapy in Necrotizing Fasciitis Reduces the Number of Surgical Debridements. Wounds, 2007. 19(3): pp. 73–78.
78. Fonseca‐Muñoz, A., et al., Clinical Study of Maggot therapy for Fournier’s Gangrene. International Wound Journal, 2020. 17(6): pp. 1642–1649, https://doi.org/10.1111/iwj.13444.
79. Berger, S., Tropical Skin Ulcers: Global Status. 2017, Los Angeles: Gideon Informatics, Inc.
80. Frimpong, M., et al., Paradoxical Reactions in Buruli Ulcer after Initiation of Antibiotic Therapy: Relationship to Bacterial Load. PLOS Neglected Tropical Diseases, 2019. 13(8): e0007689-e0007689, https://doi.org/10.1371/journal.pntd.0007689.
81. Eichelmann, K., et al., Leprosy. An update: Definition, Pathogenesis, Classification, Diagnosis, and Treatment. Actas Dermo-Sifiliograficas, 2013. 104(7): pp. 554–563, https://doi.org/10.1016/j.ad.2012.03.003.
82. Arrivillaga, J., J. Rodríguez, and M. Oviedo, Preliminary Evaluation of Maggot (Diptera: Calliphoridae) Therapy as a Potential Treatment for Leishmaniasis Ulcers. Biomédica, 2008. 28(2): pp. 305–310, https://doi.org/10.7705/biomedica.v28i2.102.
83. Cruz-Saavedra, L., et al., The Effect of Lucilia sericata- and Sarconesiopsis magellanica-derived Larval Therapy on Leishmania panamensis. Acta Tropica, 2016. 164: pp. 280–289, https://doi.org/10.1016/j.actatropica.2016.09.020.
84. Polat, E., et al., Detection of Anti-leishmanial Effect of the Lucilia sericata Larval Secretions in vitro and in vivo on Leishmania tropica: First work. Experimental Parasitology, 2012. 132(2): pp. 129–134, https://doi.org/10.1016/j.exppara.2012.06.004.
85. Sanei-Dehkordi, A., et al., Anti Leishmania Activity of Lucilia sericata and Calliphora vicina Maggots in Laboratory Models. Experimental Parasitology, 2016. 170: pp. 59–65, https://doi.org/10.1016/j.exppara.2016.08.007.
86. Stadler, F., R.Z. Shaban, and P. Tatham, Maggot Debridement Therapy in Disaster Medicine. Prehospital and Disaster Medicine, 2016. 31(1): pp. 79–84, https://doi.org/10.1017/S1049023X15005427.
87. Čičková, H., M. Kozánek, and P. Takáč, Growth and Survival of Blowfly Lucilia sericata Larvae under Simulated Wound Conditions: Implications for Maggot Debridement Therapy. Medical and Veterinary Entomology, 2015. 29(4): pp. 416–424, https://doi.org/10.1111/mve.12135.
88. Stadler, F. and P. Tatham, Drone-assisted Medicinal Maggot Distribution in Compromised Healthcare Settings, in A Complete Guide to Maggot Therapy: Clinical Practice, Therapeutic Principles, Production, Distribution, and Ethics, F. Stadler (ed.). 2022, Cambridge: Open Book Publishers, pp. 383–402, https://doi.org/10.11647/OBP.0300.18.
89. Ayello, E.A., et al., Reexamining the Literature on Terminal Ulcers, SCALE, Skin Failure, and Unavoidable Pressure Injuries. Advances in Skin & Wound Care, 2019. 32(3): pp. 109–121, https://doi.org/10.1097/01.ASW.0000553112.55505.5f.
90. Steenvoorde, P., et al., Maggot Debridement Therapy in the Palliative Setting. American Journal of Hospice & Palliative Medicine, 2007. 24(4): pp. 308–310, https://doi.org/10.1177/1049909107302300.
91. Brüggmann, D., H.R. Tinneberg, and M.T. Zygmunt, [Maggot Therapy in Gynecology]. Zentralblatt für Gynäkologie 2006. 128(5): pp. 261–265, https://doi.org/10.1055/s-2006-942121.
92. Wolters, U., et al., ASA Classification and Perioperative Variables as Predictors of Postoperative Outcome. British Journal of Anaesthesia, 1996. 77(2): Pp. 217–222, https://doi.org/10.1093/bja/77.2.217.
93. Peck, G.W. and B.C. Kirkup, Biocompatibility of Antimicrobials to Maggot Debridement Therapy: Medical Maggots Lucilia sericata (Diptera: Calliphoridae) Exhibit Tolerance to Clinical Maximum Doses of Antimicrobials. Journal of Medical Entomology, 2012. 49(5): pp. 1137–1143, https://doi.org/10.1603/ME12066.
94. Sherman, R.A., F.A. Wyle, and L. Thrupp, Effects of Seven Antibiotics on the Growth and Development of Phaenicia sericata (Diptera: Calliphoridae) Larvae. Journal of Medical Entomology, 1995. 32(5): pp. 646–649, https://doi.org/10.1093/jmedent/32.5.646.
95. Felder, J.M., et al., Increasing the Options for Management of Large and Complex Chronic Wounds with a Scalable, Closed-system Dressing for Maggot Therapy. Journal of Burn Care & Research, 2012. 33(3): pp. e169-e175, https://doi.org/10.1097/BCR.0b013e318233570d.
96. Sherman, R.A., B. Khavari, and D. Werner, Effect of Hyperbaric Oxygen on the Growth and Development of Medicinal Maggots. Undersea and Hyperbaric Medicine, 2013. 40(5): pp. 377–380.
97. Teich, S. and R.A.M. Myers, Maggot Therapy for Severe Skin Infections. Southern Medical Journal, 1986. 79(9): pp. 1153–1155.
98. Rojo, S. and S. Geraghty, Hemophilia and Maggots: From Hospital Admission to Healed Wound. Ostomy Wound Management, 2004. 50(4): pp. 30, 32, 34.
99. Sherman, R.A. and M.R. Hetzler, Maggot Therapy for Wound Care in Austere Environments. Journal of Special Operations Medicine, 2017. 17(2): pp. 154–162.
100. Grassberger, M. and C. Reiter, Effect of Temperature on Lucilia sericata (Diptera: Calliphoridae) Development with Special Reference to the Isomegalen- and Isomorphen-diagram. Forensic Science International, 2001. 120(1): pp. 32–36, https://doi.org/10.1016/S0379-0738(01)00413-3.
101. Sherman, R.A., et al., Effects of Food Storage and Handling on Blow Fly (Lucilia sericata) Eggs and Larvae. Journal of Food Science, 2006. 71(3): pp. M117-M120, https://doi.org/10.1111/j.1365-2621.2006.tb15634.x.
102. Gallagher, M.B., S. Sandhu, and R. Kimsey, Variation in Developmental Time for Geographically Distinct Populations of the Common Green Bottle Fly, Lucilia sericata (Meigen). Journal of Forensic Sciences, 2010. 55(2): pp. 438–442, https://doi.org/10.1111/j.1556-4029.2009.01285.x.
103. Richards, C.S. and M.H. Villet, Data Quality in Thermal Summation Development Models for Forensically Important Blowflies. Medical and Veterinary Entomology, 2009. 23(3): pp. 269–276, https://doi.org/10.1111/j.1365-2915.2009.00819.x.
104. Mirabzadeh, A., et al., Maggot Therapy for Wound Care in Iran: A Case Series of the First 28 Patients. Journal of Wound Care, 2017. 26(3): pp. 137–143, https://doi.org/10.12968/jowc.2017.26.3.137.
105. Lin, G., et al., Hard Times Call for Creative Solutions: Medical Improvisations at the Israel Defense Forces Field Hospital in Haiti. American Journal of Disaster Medicine, 2010. 5(3): pp. 188–192.
