8. Maggot Debridement
© 2022 Nigam & Wilson, CC BY-NC 4.0 https://doi.org/10.11647/OBP.0300.08
For non-healing wounds to progress past the inflammatory stage, it is vital that necrotic tissue is quickly and effectively removed, a treatment that is known as debridement. Maggot therapy is the treatment of wounds with living fly larvae (maggots) to remove necrotic tissue. In recent years, much progress has been made in understanding the therapeutic principles of maggot-assisted debridement. This chapter describes the physiological and biochemical principles underpinning the extraordinary ability of medicinal maggots to precisely debride highly necrotic wounds in a matter of days without the need for surgical intervention.
Introduction
Non-healing wounds containing necrotic tissue can be attributed to a variety of factors including chronic disease, vascular insufficiencies, advanced age, neurological defects, nutritional deficiencies, or local factors such as infection, pressure, and oedema [1]. In these wounds, the normal healing process stalls, typically in a chronic state of inflammation, causing a cascade of abnormal tissue responses which can generate and amplify a hostile microenvironment inside the wound. This results in accumulation of cellular debris on the wound surface and damage to surrounding tissue, leading to infection and necrosis [2, 3]. To aid the process of wound healing and allow it to progress past the inflammatory stage, it is vital that this necrotic tissue is quickly and effectively removed, a treatment that is known as debridement [4, 5].
Maggot therapy is the treatment of wounds with living fly larvae (maggots) in need of debridement. The ability of Lucilia sericata and other fly species to debride is attributed to their necrophagous nature. This is to say that their primary source of nutrition in the wild is dead and rotting tissue [6]. The larvae of L. sericata consume the necrotic tissue, and it is their voracious appetite for this material that makes them so effective in this process (Figure 8.1).
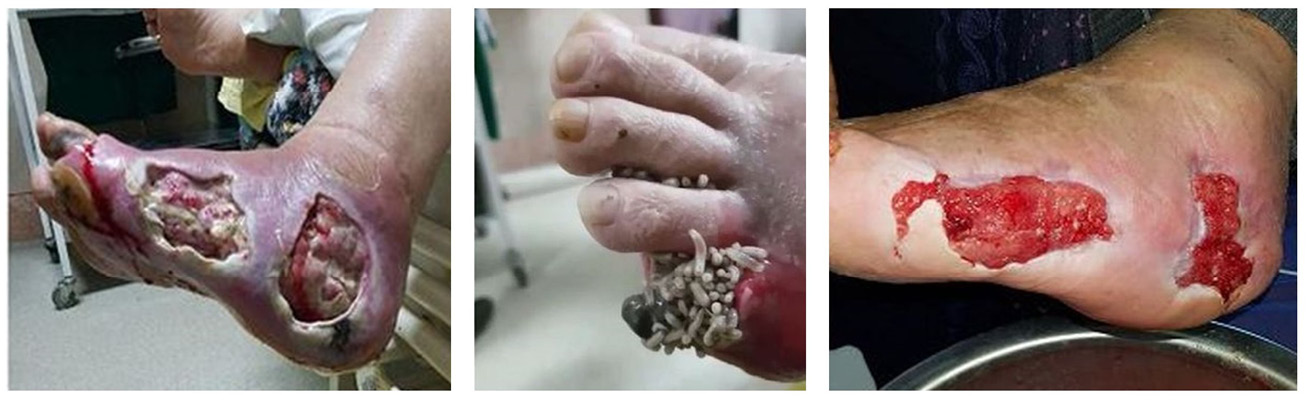
Figure 8.1 Maggot therapy with free-range larvae (Photos by Parizad et al. 2021, https://doi.org/10.1016/j.ijscr.2021.105931 [7], CC BY-NC.
Efficacy of Maggot Therapy for Wound Debridement
Since its resurgence in the USA and UK in the 1990s, a number of clinical studies have been conducted to compare the efficacy of larval therapy to conventional treatment methods in debriding chronic wounds. A systematic review of the clinical studies of larval therapy incorporated twelve comparative studies, including six randomised controlled trials, from the years 2000–2014 [8]. Based on the analysis of these twelve studies, the authors concluded that larval therapy was both more effective and more efficient in the debridement of chronic ulcers when compared with conventional treatments. They also associated larval therapy with other benefits, including quicker healing rate of chronic wounds, a shortened time to healing in ulcers, a longer antibiotic-free time period, decreased amputation risk, and similar antibiotic usage compared with conventional therapies [8]. A subsequent review of clinical studies of maggot therapy from 2000–2015 corroborated the positive debridement results, showing a significantly higher rate of successful debridement compared with conventional treatments and a significantly faster time for wounds to heal [9].
The Feeding Process and Maggot Enzymatic Action
In the wild, carrion is a nutritive but ephemeral resource, and there is intense competition among arthropods to acquire these resources before they are depleted. Therefore, like many other species of Calliphoridae, L. sericata larvae have adapted some particularly effective mechanisms to satisfy their nutritional requirements and ensure rapid development [10–12]. Chapter 7 of this book explains the natural history of calliphorid flies, including their feeding strategies [6]. The primary feeding mechanism of larvae is extracorporeal digestion, a process that was first demonstrated in the 1930s [13]. The food is digested externally by larval excretions/secretions which contain a potent mixture of proteolytic enzymes, deoxyribonuclease, ammonia, and antimicrobial substances. The movement of the larvae over the food then facilitates the penetration of these secretions into the necrotic tissue, causing it to break down and liquefy into a nutrient-rich fluid that the larvae subsequently ingest [14, 15]. This method of feeding is particularly well-suited to individuals feeding together in large populations such as those seen in blowfly larvae as it enables them to share and combine the enzymatic secretions they release, allowing them to feed more efficiently [16, 17]. Additionally, the secretions produced by maggots work to increase pH in the wound environment, making it more conducive to the action of the proteolytic enzymes [15, 18, 19]. It is also believed that the secretions help to irrigate bacteria from the wound environment [20] as discussed in more detail in Chapter 9 [21]. These initial debridement and disinfection processes are vital for wound healing to proceed [22].
As well as the enzymatic action, it is also thought that debridement in larval therapy is aided by the physical action of the larvae themselves. Each larva possesses a set of mouth hooks which they use to scrape and tear at the surface tissue, and it is thought that the most significant function of this action is to facilitate the penetration of their secretions into the tissue, thereby making it easier to break down [23, 24]. The mouth hooks are also used for locomotion as they help to pull the body forward when maggots crawl [25]. Additionally, larvae seek out areas containing necrotic tissue and thus ensure that debridement activity is focussed on the areas of greatest need [26].
Identification of Maggot Enzymes in Debridement
The enzymes that are contained in larval secretions are key to wound debridement. Therefore, identifying those enzymes responsible would be very helpful in further understanding the action of larvae in the chronic wound. Initial work looked at characterising the types of enzymes at work. Using class specific substrates and inhibitors, Chambers and Woodrow [15] identified three classes of proteolytic enzymes at work in the process of larval debridement: serine proteases, aspartyl proteases, and metalloproteases. Of these enzymes, the most significant activity was attributed to serine proteases of two different sub-classes, trypsin-like and chymotrypsin-like. Larvae also provide the optimal conditions for the serine and metalloproteases to act within the wound, secreting ammonia to increase the pH in the wound bed and thus allowing activation of trypsin-like proteases (which have a role in cell proliferation, cytokine secretion and ultimately wound healing) and chymotrypsin-like proteases that degrade the extracellular matrix components laminin, fibronectin and collagen types I and III [15].
Serine proteases were also subsequently identified in larval secretions in a separate study, indicating the role of these enzymes in wound debridement [27]. A recent, novel investigation using a physical model corroborated this idea. The work involved the incorporation of trypsin inhibitors in the feed of larvae so that enzymes that were produced by the larvae were inhibited. The result of this was a significant decrease in consumption rates and a severe stunting of larval growth [28]. This work confirmed that larvae did indeed utilise trypsin proteases for extracorporeal digestions and subsequent consumption of dead tissue. Trypsin inhibitors have also been successfully incorporated into the diets of other insect species, which has resulted in similarly detrimental effects on both feeding and development [29–32]. This means that the use of serine proteases in digestion is not unique to L. sericata.
There has also been work to identify the individual enzymes at work. It had been demonstrated that a chymotrypsin-like serine protease was able to degrade extracellular matrix components in wounds and aid fibroblast migration through this action. The three major components in extracellular matrix that were degraded by this chymotrypsin were fibronectin, laminin and collagen [15]. This chymotrypsin was subsequently identified and characterised from L. sericata larvae and a recombinant form (named “chymotrypsin I”) was shown to be effective in degrading venous leg wound eschar ex vivo [33–35]. Utilising various techniques including 2D gel electrophoresis and the monitoring of the release of 7-amino-4-methyl coumarin from the chymotrypsin substrate, it was concluded that the active recombinant chymotrypsin I degraded the wound eschar more efficiently than commercially available chymotrypsin from bovine and human sources [35].
Additionally, chymotrypsin I was found not to be restrained by endogenous inhibitors that were found in high concentrations within wound slough (α-1-antichymotrypsin and α-1-antitrypsin), indicating a means by which the activity of the enzyme is able to survive within the wound and contribute to debridement [36]. The protease is, however, inhibited by α-2-macroglobulin which is significant because it is a macromolecule found in blood plasma which in turn is abundant in healthy and blood-perfused tissue. It is hypothesised that the specificity of L. sericata larvae for digesting only necrotic tissue may be explained by this inhibitory effect of α-2-macroglobulin [37].
Larval secretions have also been found to contain deoxyribonuclease, capable of degrading genomic and extracellular bacterial DNA, as well as DNA from wound slough, indicating a potential to aid in wound debridement and inhibition of the growth of bacteria and biofilm [14]. In another study, a complementary DNA library was constructed from the salivary glands of medicinal larvae and five full-length and several incomplete complimentary DNAs encoding for serine proteases were identified [38]. Further work to characterise the active enzymes in larval secretions resulted in the identification of a chymotrypsin-like serine protease known as “Jonahm” which was found to digest certain extracellular matrix components normally present in the chronic wound environment [39]. However, this enzyme was found not to digest certain other components, suggesting that it is a complex mix of enzymes in larval secretions that facilitate debridement. This was confirmed in another study where a comprehensive analysis of the proteolytic enzymes released from larval tissues—including the salivary glands, crop, and gut—identified hundreds of clusters representing five classes of enzyme: aspartic, cysteine, threonine, serine, and metallopeptidases [40]. Serine peptidases represented the largest group of peptidases identified from L. sericata, and in addition to the previously characterised proteases, dozens more were identified with roles in digestion, immunity, blood coagulation, and others whose roles are still unclear [40].
The significance of uncovering the therapeutically active substances in larval secretions lies not only in gaining a greater understanding of how larval therapy works, but also in aiding the possible development of new treatments. Whilst the efficacy of larval therapy is well proven, its widespread adoption is limited by practical problems associated with using live organisms. These include a short shelf-life, the need for an advanced logistics network to allow for express delivery, and the need for extra training to ensure that dressings are applied and maintained correctly [33]. By uncovering the active components of larvae secretions, it is envisioned that these molecules could be synthesised and then delivered by a suitable device or mechanism without having to use live larvae.
Summary
Maggot therapy is made possible by the extraordinary life-history characteristics of medicinal flies, and by the feeding behaviour and physiology of their larvae. Adapted to the exploitation of ephemeral cadavers, they have evolved the capacity to rapidly consume decaying flesh and complete their larval growth in just a few days. A cocktail of digestive enzymes is secreted to dissolve only necrotic tissue before the nutrient-rich liquid digest is consumed. When such flies colonise the wound of a living human or animal (myiasis) and conditions are conducive, then their gentle, selective, and highly efficient feeding behaviour brings about rapid debridement of the wound.
This therapeutic benefit of myiasis has been recognised since ancient times and adopted by tribal healers and later by modern medicine. In recent years, much progress has been made in understanding the therapeutic principles of maggot-assisted debridement. The digestive secretions of course play a critical role. Now the race is on to isolate and synthetically produce therapeutic compounds for wound care drug development. However, the use of pharmaceuticals derived from maggot products may have limitations. For example, the use of individual products for debridement will likely preclude the other benefits associated with treatment using live larvae such as disinfection and wound healing, and may therefore limit the effectiveness of the treatment. Additionally, considering that secretions consist of a complex mixture of enzymes [40], one can argue that the efficacy of maggot therapy relies on this complex mix of substances that are produced by the live larvae and then deposited into the wound. In other words, the substitution of this mix of substances for a single, cost-effective pharmaceutical, or even a combination of pharmaceuticals, may not be possible. How medicinal maggots control infection and bring about wound healing is discussed in Chapters 9 and 10 of this book [21, 41]. There has been some success in producing recombinant forms of maggot-derived digestive enzymes and incorporating them into a delivery device [33, 35, 39], but more work is needed to determine which enzymes are effective, how their efficacy compares against live larvae, and how they can be produced reliably at a large scale for a reasonable cost.
References
1. Fonder, M.A., et al., Treating the Chronic Wound: A Practical Approach to the Care of Nonhealing Wounds and Wound Care Dressings. Journal of the American Academy of Dermatology, 2008. 58(2): pp. 185–206, https://doi.org/10.1016/j.jaad.2007.08.048.
2. Eming, S.A., T. Krieg, and J.M. Davidson, Inflammation in Wound Repair: Molecular and Cellular Mechanisms. Journal of Investigative Dermatology, 2007. 127(3): pp. 514–525, https://doi.org/10.1038/sj.jid.5700701.
3. Zhao, R., et al., Inflammation in Chronic Wounds. International Journal of Molecular Sciences, 2016. 17(12): pp. 2085–2085, https://doi.org/10.3390/ijms17122085.
4. Wilcox, J.R., M.J. Carter, and S. Covington, Frequency of Debridements and Time to Heal: A Retrospective Cohort Study of 312,744 wounds. JAMA Dermatology, 2013. 149(9): pp. 1050–1058, https://doi.org/10.1001/jamadermatol.2013.4960.
5. Wolcott, R.D., J.P. Kennedy, and S.E. Dowd, Regular Debridement is the Main Tool for Maintaining a Healthy Wound Bed in Most Chronic Wounds. Journal of Wound Care, 2009. 18(2): pp. 54–56, https://doi.org/10.12968/jowc.2009.18.2.38743.
6. Harvey, M., The Natural History of Medicinal Flies, in A Complete Guide to Maggot Therapy: Clinical Practice, Therapeutic Principles, Production, Distribution, and Ethics, F. Stadler (ed.). 2022, Cambridge: Open Book Publishers, pp. 121–142, https://doi.org/10.11647/OBP.0300.07.
7. Parizad, N., K. Hajimohammadi, and R. Goli, Surgical Debridement, Maggot Therapy, Negative Pressure Wound Therapy, and Silver Foam Dressing Revive Hope for Patients with Diabetic Foot Ulcer: A Case Report. International Journal of Surgery Case Reports, 2021. 82: p. 105931, https://doi.org/10.1016/j.ijscr.2021.105931.
8. Sun, X., et al., A Systematic Review of Maggot Debridement Therapy for Chronically Infected Wounds and Ulcers. International Journal of Infectious Diseases, 2014. 25: pp. 32–37, https://doi.org/10.1016/j.ijid.2014.03.1397.
9. Siribumrungwong, B., C. Wilasrusmee, and K. Rerkasem, Maggot Therapy in Angiopathic Leg Ulcers: A Systematic Review and Meta-analysis. The International Journal of Lower Extremity Wounds, 2018. 17(4): pp. 227–235, https://doi.org/10.1177/1534734618816882.
10. Hanski, I., Carrion Fly Community Dynamics: Patchiness, Seasonality and Coexistence. Ecological Entomology, 1987. 12(3): pp. 257–266, https://doi.org/10.1111/j.1365-2311.1987.tb01004.x.
11. Rivers, D.B., C. Thompson, and R. Brogan, Physiological Trade-offs of Forming Maggot Masses by Necrophagous Flies on Vertebrate Carrion. Bulletin of Entomological Research, 2011. 101(05): pp. 599–611, https://doi.org/10.1017/S0007485311000241.
12. von Zuben, C.J., F.J. von Zuben, and W.A.C. Godoy, Larval Competition for Patchy Resources in Chrysomya Megacephala (Dipt., Calliphoridae): Implications of the Spatial Distribution of Immatures. Journal of Applied Entomology, 2001. 125(9–10): pp. 537–541, https://doi.org/10.1046/j.1439-0418.2001.00586.x.
13. Hobson, R.P., On an Enzyme from Blow-fly Larvae Lucilia sericata which Digests Collagen in Alkaline Solution. Biochemical Journal, 1931. 25(5): pp. 1458–1463, https://doi.org/10.1042/bj0251458.
14. Brown, A., et al., Blow Fly Lucilia sericata Nuclease Digests DNA Associated with Wound Slough/Eschar and with Pseudomonas aeruginosa Biofilm. Medical and Veterinary Entomology, 2012. 26(4): pp. 432–439, https://doi.org/10.1111/j.1365-2915.2012.01029.x.
15. Chambers, L., et al., Degradation of Extracellular Matrix Components by Defined Proteinases from the Greenbottle Larva Lucilia sericata Used for the Clinical Debridement of Non-healing Wounds. British Journal of Dermatology, 2003. 148(1): pp. 14–23, https://doi.org/10.1046/j.1365-2133.2003.04935.x.
16. dos Reis, S.F., C.J. von Zuben, and W.A.C. Godoy, Larval Aggregation and Competition for Food in Experimental Populations of Chrysomya Putoria (Wied.) and Cochliomyia Macellaria (F.) (Dipt., Calliphoridae). Journal of Applied Entomology, 1999. 123(8): pp. 485–489, https://doi.org/10.1046/j.1439-0418.1999.00397.x.
17. Ireland, S. and B. Turner, The Effects of Larval Crowding and Food Type on the Size and Development of the Blowfly, Calliphora Vomitoria. Forensic Science International, 2006. 159(2–3): pp. 175–181, https://doi.org/10.1016/j.forsciint.2005.07.018.
18. Mumcuoglu, K.Y., et al., Destruction of Bacteria in the Digestive Tract of the Maggot of Lucilia sericata (Diptera: Calliphoridae). Journal of Medical Entomology, 2001. 38(2): pp. 161–166, https://doi.org/10.1603/0022-2585-38.2.161.
19. Thomas, S., Maggot Therapy. 2010, Medetec: Cardiff. pp. 563–632.
20. Mumcuoglu, K.Y., et al., Maggot Therapy for the Treatment of Intractable Wounds. International Journal of Dermatology, 1999. 38(8): pp. 623–627, https://doi.org/10.1046/j.1365-4362.1999.00770.x.
21. Nigam, Y. and M.R. Wilson, The Antimicrobial Activity of Medicinal Maggots, in A Complete Guide to Maggot Therapy: Clinical Practice, Therapeutic Principles, Production, Distribution, and Ethics, F. Stadler (ed.). 2022, Cambridge: Open Book Publishers, pp. 153–174, https://doi.org/10.11647/OBP.0300.09.
22. Gailit, J. and R.A.F. Clark, Wound Repair in the Context of Extracellular Matrix. Current Opinion in Cell Biology, 1994. 6(5): pp. 717–725, https://doi.org/10.1016/0955-0674(94)90099-X.
23. Barnard, D.R., Skeletal-muscular Mechanisms of the Larva of Lucilia sericata (Meigen) in Relation to Feeding Habit (Diptera: Calliphoridae). Pan-Pacific Entomologist, 1977. 53(3): pp. 223–229.
24. Thomas, S., et al., The Effect of Containment on the Properties of Sterile Maggots. British Journal of Nursing, 2002. 11(Sup2): pp. S21-S28, https://doi.org/10.12968/bjon.2002.11.Sup2.10294.
25. Sherman, R.A., et al., Maggot Therapy, in Biotherapy — History, Principles and Practice, M. Grassberger, et al. (eds). 2013, Springer: Dordrecht; New York. pp. 5–29.
26. Church, J.C.T., Larva Therapy in Modern Wound Care: A Review. Primary Intention, 1999. 7(2): pp. 63–68.
27. Schmidtchen, A., et al., Detection of Serine Proteases Secreted by Lucilia sericata in vitro and during Treatment of a Chronic Leg Ulcer. Acta Dermato-Venereologica, 2003. 83(4): pp. 310–311, https://doi.org/10.1080/00015550310016689.
28. Wilson, M.R., et al., The Impacts of Larval Density and Protease Inhibition on Feeding in Medicinal Larvae of the Greenbottle Fly Lucilia sericata. Medical and Veterinary Entomology, 2016. 30(1): pp. 1–7, https://doi.org/10.1111/mve.12138.
29. Burgess, E.P.J., L.A. Malone, and J.T. Christeller, Effects of Two Proteinase Inhibitors on the Digestive Enzymes and Survival of Honey Bees Apis Mellifera. Journal of Insect Physiology, 1996. 42(9): pp. 823–828, https://doi.org/10.1016/0022-1910(96)00045-5.
30. Franco, O.L., et al., Effects of Soybean Kunitz Trypsin Inhibitor on the Cotton Boll Weevil Anthonomus Grandis. Phytochemistry, 2004. 65(1): pp. 81–89, https://doi.org/10.1016/j.phytochem.2003.09.010.
31. Johnston, K.A., J.A. Gatehouse, and J.H. Anstee, Effects of Soybean Protease Inhibitors on the Growth and Development of Larval Helicoverpa Armigera. Journal of Insect Physiology, 1993. 39(8): pp. 657–664, https://doi.org/10.1016/0022-1910(93)90071-X.
32. McManus, M.T. and E.P.J. Burgess, Effects of the Soybean (Kunitz) Trypsin Inhibitor on Growth and Digestive Proteases of Larvae of Spodoptera Litura. Journal of Insect Physiology, 1995. 41(9): pp. 731–738, https://doi.org/10.1016/0022-1910(95)00043-T.
33. Britland, S., et al., Recombinant <i>Lucilia sericata</i> Chymotrypsin in a Topical Hydrogel Formulation Degrades Human Wound Eschar ex vivo. Biotechnology Progress, 2011. 27(3): pp. 870–874, https://doi.org/10.1002/btpr.587.
34. Pritchard, D.I. and A.P. Brown, Degradation of MSCRAMM Target Macromolecules in VLU Slough by Lucilia sericata Chymotrypsin 1 (ISP) Persists in the Presence of Tissue Gelatinase Activity. International Wound Journal, 2015. 12(4): pp. 414–421, https://doi.org/10.1111/iwj.12124.
35. Telford, G., et al., Degradation of Eschar from Venous Leg Ulcers Using a Recombinant Chymotrypsin from Lucilia sericata. British Journal of Dermatology, 2010. 163(3): pp. 523–531, https://doi.org/10.1111/j.1365-2133.2010.09854.x.
36. Telford, G., et al., Maggot Chymotrypsin I from Lucilia sericata Is Resistant to Endogenous Wound Protease Inhibitors. British Journal of Dermatology, 2011. 164(1): pp. 192–196, https://doi.org/10.1111/j.1365-2133.2010.10081.x.
37. Pritchard, D.I., et al., TIME Management by Medicinal Larvae. International Wound Journal, 2016. 13(4): pp. 475–484, https://doi.org/10.1111/iwj.12457.
38. Valachova, I., et al., Identification and Characterisation of Different Proteases in Lucilia sericata Medicinal Maggots Involved in Maggot Debridement Therapy. Journal of Applied Biomedicine, 2014. 12(3): pp. 171–177, https://doi.org/10.1016/j.jab.2014.01.001.
39. Pöppel, A., et al., A Jonah-like Chymotrypsin from the Therapeutic Maggot Lucilia sericata Plays a Role in Wound Debridement and Coagulation. Insect Biochemistry and Molecular Biology, 2016. 70: pp. 138–147, https://doi.org/10.1016/j.ibmb.2015.11.012.
40. Franta, Z., et al., Next Generation Sequencing Identifies Five Major Classes of Potentially Therapeutic Enzymes Secreted by Lucilia sericata Medical Maggots. BioMed Research International, 2016. 2016: pp. 1–27, https://doi.org/10.1155/2016/8285428.
41. Nigam, Y. and M.R. Wilson, Maggot-assisted Wound Healing, in A Complete Guide to Maggot Therapy: Clinical Practice, Therapeutic Principles, Production, Distribution, and Ethics, F. Stadler (ed.). 2022, Cambridge: Open Book Publishers, pp. 175–194, https://doi.org/10.11647/OBP.0300.10.
